Hyperthyroidism symptoms pregnancy
HYPERthyroidism in Pregnancy | InfantRisk Center
Thyroid diseases are among the most common endocrine disorders encountered during pregnancy. The prevalence of hyperthyroidism (overactive thyroid) during pregnancy ranges from 0.05 to 0.2%. The most common causes of hyperthyroidism during pregnancy are Graves’ diseaseand hCG-mediated hyperthyroidism such as gestational transient hyperthyroidism, hyperemesis gravidarum (intractable nausea and vomiting), and gestational trophoblastic disease. Other causes of hyperthyroidism include toxic adenoma, subacute thyroiditis, and induced thyroxine overdose.1, 2
Poorly controlled hyperthyroidism during pregnancy has been associated with many maternal and fetal risks. The most common complication of hyperthyroidism in pregnant mothers is preeclampsia. Other reported complications include miscarriage, pre-term labor, heart failure, and stillbirth. Fetal risks include low-birth-weight infants, induced fetal hypothyroidism, and fetal thyrotoxicosis. 1, 3, 4
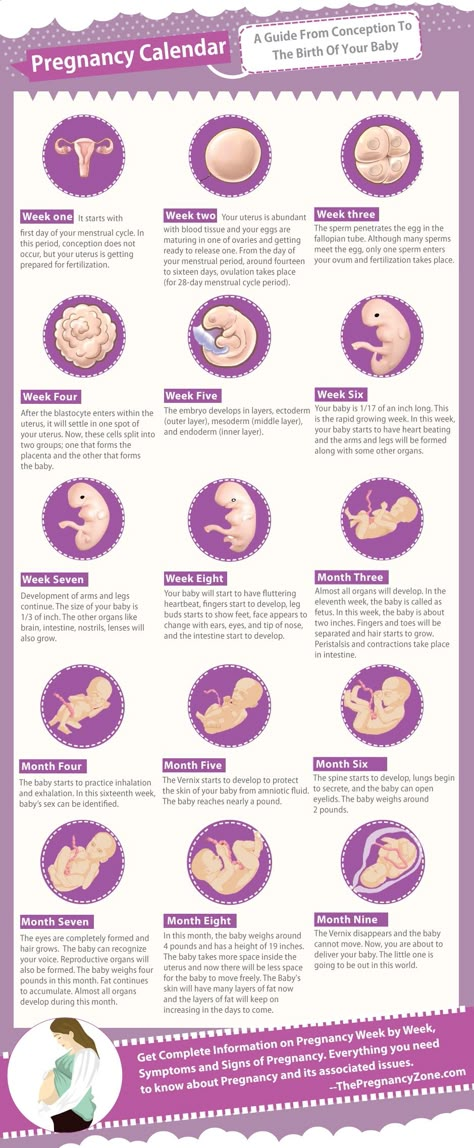
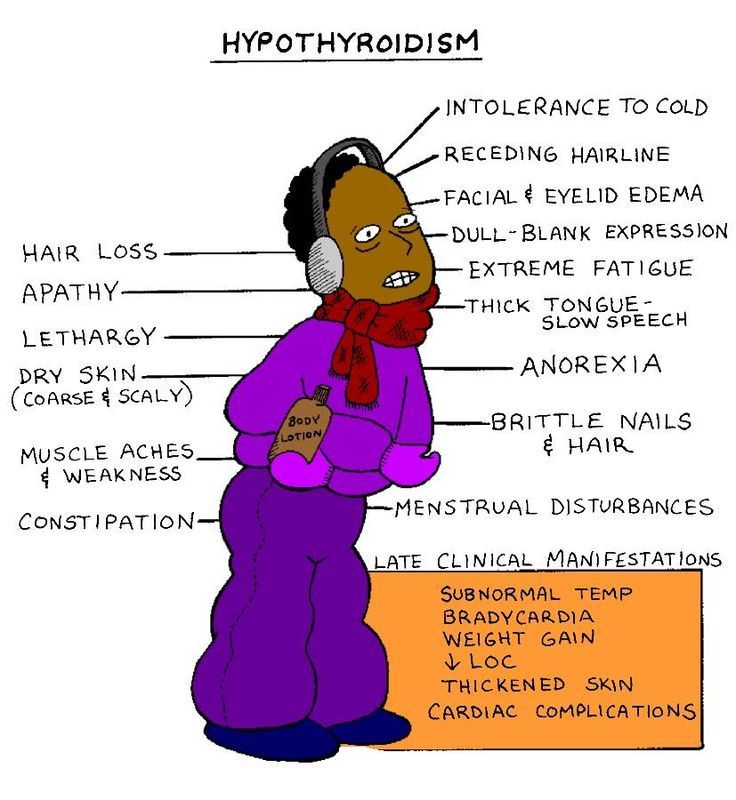
The signs and symptoms of hyperthyroidism during normal pregnancy include heat intolerance, fatigue, anxiety, increase perspiration, tachycardia (increase heart rate), hand tremor, severe nausea and vomiting, and weight loss despite a normal or increased appetite.1 Specific findings such as goiter and exophthalmos (protruding eyes) suggest Graves' hyperthyroidism. Other manifestations such as confusion and abnormally increased metabolic rate characterize a very rare life-threatening complication known as thyroid storm which can be precipitated by labor, infection, cesarean section, or gestational trophoblastic disease.5
The diagnosis of hyperthyroidism during pregnancy is based upon clinical manifestations and laboratory findings. Values of < 0.1 mU/L of serum TSH and elevated thyroid hormone levels (serum free T4 and/or free T3 or total T4 and/or total T3) that exceed the normal range for pregnancy.5 In addition, the presence of TSH receptor antibodies is diagnostic of Graves' disease. 6 Radioactive iodine is contraindicated in the diagnosis of hyperthyroidism during pregnancy because it crosses the placenta and induces fetal hypothyroidism and increased risk of thyroid carcinoma.7
6 Radioactive iodine is contraindicated in the diagnosis of hyperthyroidism during pregnancy because it crosses the placenta and induces fetal hypothyroidism and increased risk of thyroid carcinoma.7
Treatment options for pregnant women with overt hyperthyroidism are limited because therapy may be harmful to the fetus. However, a good fetal and maternal outcome depends upon controlling the mother's hyperthyroidism.7 Especially, aggressive treatment for thyroid storm is critical to the patient’s survival. According to the Endocrine Society and the American Thyroid Association (ATA), the goals of treatment are to provide symptomatic relief, to keep fT4 (free T4) levels in the upper third of normal (normal range of fT4 : 0.8-1.2 ng/dL for first trimester, 0.6-1.0 ng/dL for second trimester, and 0.5-0.8 ng/dL for third trimester), and to rapidly achieve and maintain euthyroidism (normal thyroid state) with the lowest effective antithyroid doses.1
Symptomatic women and/or moderate to severe overt hyperthyroid women, which are due to thyroid storm, Graves’ disease, toxic adenoma, toxic multinodular goiter, or gestational trophoblastic disease, all require therapy treatment.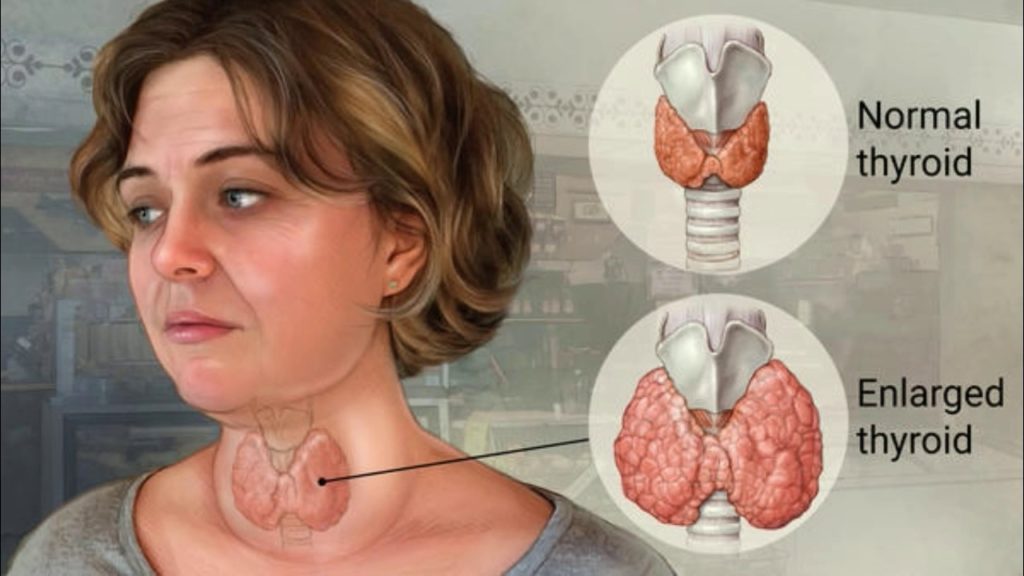 However, asymptomatic mothers with subclinical and/or mild hyperthyroidism such as hCG-mediated hyperthyroidism may be followed with no treatment. These women may need their TSH and free T4 measured every four to six weeks.7
However, asymptomatic mothers with subclinical and/or mild hyperthyroidism such as hCG-mediated hyperthyroidism may be followed with no treatment. These women may need their TSH and free T4 measured every four to six weeks.7
The recommended treatment for hyperthyroidism in the first trimester of pregnancy is propylthiouracil (PTU) ≤ 50 mg twice daily, then switching to ≤ 5 to 10 mg daily of methimazole at the start of second trimester. Higher doses can result in fetal goiter and hypothyroidism. This requires assessment of free T4 (and/or total T4) frequently with appropriate adjustment of medication.7
The Endocrine Society and ATA suggest that radioiodine therapy is absolutely contraindicated in pregnant women with hyperthyroidism. They also do not recommend nonradioactive iodine for long-term use because chronic use can cause fetal goiter. However, they do suggest thyroidectomy in hyperthyroid women during pregnancy when thioamides (thyroid drugs) are not tolerated due to allergy or blood disorders such as agranulocytosis. Such women should be treated with a short course (7 to 10 days) of beta blockers and iodine in preparation for thyroidectomy.7 Beta blockers can be used for symptomatic relief in severe cases but only short period of few weeks. However, used for prolonged periods of time, it can cause intrauterine growth restriction and hypoglycemia.1
Such women should be treated with a short course (7 to 10 days) of beta blockers and iodine in preparation for thyroidectomy.7 Beta blockers can be used for symptomatic relief in severe cases but only short period of few weeks. However, used for prolonged periods of time, it can cause intrauterine growth restriction and hypoglycemia.1
Lylla Ngo, M.D
Thomas Hale, Ph.D
Infantrisk Center
References:
- DeCherney A, Nathan L, Laufer N, Roman A. Current diagnosis & treatment : obstetrics & gynecology. 11th ed. ed. New York: McGraw-Hill/Medical; 2013.
- Ross D. Overview of thyroid disease in pregnancy. UpToDate; 2014.
- Luewan S, Chakkabut P, Tongsong T. Outcomes of pregnancy complicated with hyperthyroidism: a cohort study. Arch Gynecol Obstet. Feb 2011;283(2):243-247.
- Refetoff S, Weiss R, Grasberger H. Resistance of thyrotropin and thyrotropin releasing hormone. UpToDate; 2014.

- Ross D. Hyperthyroidism during pregnancy: Clinical manifestations, diagnosis, and causes. UpToDate; 2012.
- Davies T. Pathogenesis of Grave's disease. UpToDate; 2013.
- Ross D. Hyperthyroidism during pregnancy: Treatment. UpToDate; 2014.
Thyroid Disease & Pregnancy | NIDDK
On this page:
- What role do thyroid hormones play in pregnancy?
- Hyperthyroidism in Pregnancy
- Hypothyroidism in Pregnancy
- Postpartum Thyroiditis
- Is it safe to breastfeed while I’m taking beta-blockers, thyroid hormone, or antithyroid medicines?
- Thyroid Disease and Eating During Pregnancy
- Clinical Trials
Thyroid disease is a group of disorders that affects the thyroid gland. The thyroid is a small, butterfly-shaped gland in the front of your neck that makes thyroid hormones. Thyroid hormones control how your body uses energy, so they affect the way nearly every organ in your body works—even the way your heart beats.
Sometimes the thyroid makes too much or too little of these hormones. Too much thyroid hormone is called hyperthyroidism and can cause many of your body’s functions to speed up. “Hyper” means the thyroid is overactive. Learn more about hyperthyroidism in pregnancy. Too little thyroid hormone is called hypothyroidism and can cause many of your body’s functions to slow down. “Hypo” means the thyroid is underactive. Learn more about hypothyroidism in pregnancy.
If you have thyroid problems, you can still have a healthy pregnancy and protect your baby’s health by having regular thyroid function tests and taking any medicines that your doctor prescribes.
What role do thyroid hormones play in pregnancy?
Thyroid hormones are crucial for normal development of your baby’s brain and nervous system. During the first trimester—the first 3 months of pregnancy—your baby depends on your supply of thyroid hormone, which comes through the placenta. At around 12 weeks, your baby’s thyroid starts to work on its own, but it doesn’t make enough thyroid hormone until 18 to 20 weeks of pregnancy.
At around 12 weeks, your baby’s thyroid starts to work on its own, but it doesn’t make enough thyroid hormone until 18 to 20 weeks of pregnancy.
Two pregnancy-related hormones—human chorionic gonadotropin (hCG) and estrogen—cause higher measured thyroid hormone levels in your blood. The thyroid enlarges slightly in healthy women during pregnancy, but usually not enough for a health care professional to feel during a physical exam.
Thyroid problems can be hard to diagnose in pregnancy due to higher levels of thyroid hormones and other symptoms that occur in both pregnancy and thyroid disorders. Some symptoms of hyperthyroidism or hypothyroidism are easier to spot and may prompt your doctor to test you for these thyroid diseases.
Another type of thyroid disease, postpartum thyroiditis, can occur after your baby is born.
Hyperthyroidism in Pregnancy
What are the symptoms of hyperthyroidism in pregnancy?
Some signs and symptoms of hyperthyroidism often occur in normal pregnancies, including faster heart rate, trouble dealing with heat, and tiredness.
Other signs and symptoms can suggest hyperthyroidism:
- fast and irregular heartbeat
- shaky hands
- unexplained weight loss or failure to have normal pregnancy weight gain
What causes hyperthyroidism in pregnancy?
Hyperthyroidism in pregnancy is usually caused by Graves’ disease and occurs in 1 to 4 of every 1,000 pregnancies in the United States.1 Graves’ disease is an autoimmune disorder. With this disease, your immune system makes antibodies that cause the thyroid to make too much thyroid hormone. This antibody is called thyroid stimulating immunoglobulin, or TSI.
Graves’ disease may first appear during pregnancy. However, if you already have Graves’ disease, your symptoms could improve in your second and third trimesters. Some parts of your immune system are less active later in pregnancy so your immune system makes less TSI. This may be why symptoms improve. Graves’ disease often gets worse again in the first few months after your baby is born, when TSI levels go up again. If you have Graves’ disease, your doctor will most likely test your thyroid function monthly throughout your pregnancy and may need to treat your hyperthyroidism.1 Thyroid hormone levels that are too high can harm your health and your baby’s.
If you have Graves’ disease, your doctor will most likely test your thyroid function monthly throughout your pregnancy and may need to treat your hyperthyroidism.1 Thyroid hormone levels that are too high can harm your health and your baby’s.
Rarely, hyperthyroidism in pregnancy is linked to hyperemesis gravidarum—severe nausea and vomiting that can lead to weight loss and dehydration. Experts believe this severe nausea and vomiting is caused by high levels of hCG early in pregnancy. High hCG levels can cause the thyroid to make too much thyroid hormone. This type of hyperthyroidism usually goes away during the second half of pregnancy.
Less often, one or more nodules, or lumps in your thyroid, make too much thyroid hormone.
How can hyperthyroidism affect me and my baby?
Untreated hyperthyroidism during pregnancy can lead to
- miscarriage
- premature birth
- low birthweight
- preeclampsia—a dangerous rise in blood pressure in late pregnancy
- thyroid storm—a sudden, severe worsening of symptoms
- congestive heart failure
Rarely, Graves’ disease may also affect a baby’s thyroid, causing it to make too much thyroid hormone. Even if your hyperthyroidism was cured by radioactive iodine treatment to destroy thyroid cells or surgery to remove your thyroid, your body still makes the TSI antibody. When levels of this antibody are high, TSI may travel to your baby’s bloodstream. Just as TSI caused your own thyroid to make too much thyroid hormone, it can also cause your baby’s thyroid to make too much.
Even if your hyperthyroidism was cured by radioactive iodine treatment to destroy thyroid cells or surgery to remove your thyroid, your body still makes the TSI antibody. When levels of this antibody are high, TSI may travel to your baby’s bloodstream. Just as TSI caused your own thyroid to make too much thyroid hormone, it can also cause your baby’s thyroid to make too much.
Tell your doctor if you’ve had surgery or radioactive iodine treatment for Graves’ disease so he or she can check your TSI levels. If they are very high, your doctor will monitor your baby for thyroid-related problems later in your pregnancy.
Tell your doctor if you’ve had surgery or radioactive iodine treatment for Graves’ disease.An overactive thyroid in a newborn can lead to
- a fast heart rate, which can lead to heart failure
- early closing of the soft spot in the baby’s skull
- poor weight gain
- irritability
Sometimes an enlarged thyroid can press against your baby’s windpipe and make it hard for your baby to breathe. If you have Graves’ disease, your health care team should closely monitor you and your newborn.
If you have Graves’ disease, your health care team should closely monitor you and your newborn.
How do doctors diagnose hyperthyroidism in pregnancy?
Your doctor will review your symptoms and do some blood tests to measure your thyroid hormone levels. Your doctor may also look for antibodies in your blood to see if Graves’ disease is causing your hyperthyroidism. Learn more about thyroid tests and what the results mean.
How do doctors treat hyperthyroidism during pregnancy?
If you have mild hyperthyroidism during pregnancy, you probably won’t need treatment. If your hyperthyroidism is linked to hyperemesis gravidarum, you only need treatment for vomiting and dehydration.
If your hyperthyroidism is more severe, your doctor may prescribe antithyroid medicines, which cause your thyroid to make less thyroid hormone. This treatment prevents too much of your thyroid hormone from getting into your baby’s bloodstream. You may want to see a specialist, such as an endocrinologist or expert in maternal-fetal medicine, who can carefully monitor your baby to make sure you’re getting the right dose.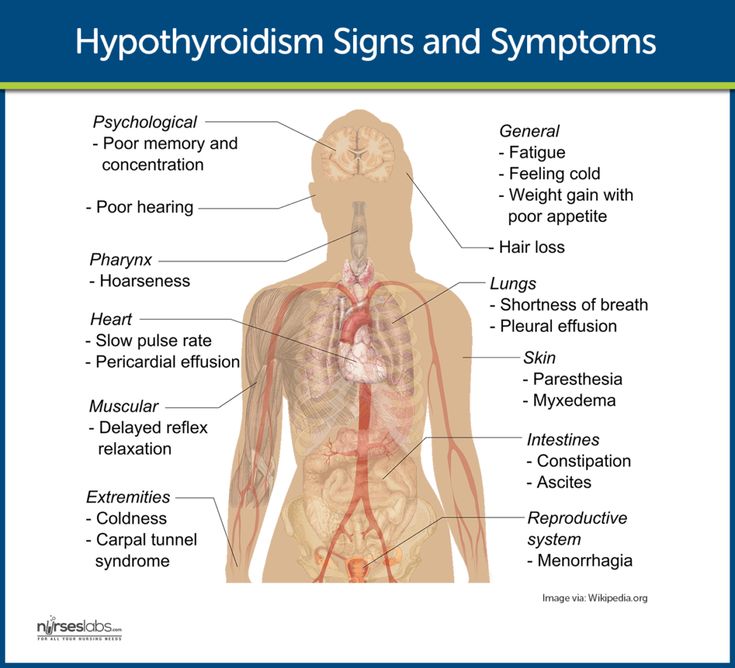
Doctors most often treat pregnant women with the antithyroid medicine propylthiouracil (PTU) during the first 3 months of pregnancy. Another type of antithyroid medicine, methimazole, is easier to take and has fewer side effects, but is slightly more likely to cause serious birth defects than PTU. Birth defects with either type of medicine are rare. Sometimes doctors switch to methimazole after the first trimester of pregnancy. Some women no longer need antithyroid medicine in the third trimester.
Small amounts of antithyroid medicine move into the baby’s bloodstream and lower the amount of thyroid hormone the baby makes. If you take antithyroid medicine, your doctor will prescribe the lowest possible dose to avoid hypothyroidism in your baby but enough to treat the high thyroid hormone levels that can also affect your baby.
Antithyroid medicines can cause side effects in some people, including
- allergic reactions such as rashes and itching
- rarely, a decrease in the number of white blood cells in the body, which can make it harder for your body to fight infection
- liver failure, in rare cases
Stop your antithyroid medicine and call your doctor right away if you develop any of these symptoms while taking antithyroid medicines:
- yellowing of your skin or the whites of your eyes, called jaundice
- dull pain in your abdomen
- constant sore throat
- fever
If you don’t hear back from your doctor the same day, you should go to the nearest emergency room.
You should also contact your doctor if any of these symptoms develop for the first time while you’re taking antithyroid medicines:
- increased tiredness or weakness
- loss of appetite
- skin rash or itching
- easy bruising
If you are allergic to or have severe side effects from antithyroid medicines, your doctor may consider surgery to remove part or most of your thyroid gland. The best time for thyroid surgery during pregnancy is in the second trimester.
Radioactive iodine treatment is not an option for pregnant women because it can damage the baby’s thyroid gland.
Hypothyroidism in Pregnancy
What are the symptoms of hypothyroidism in pregnancy?
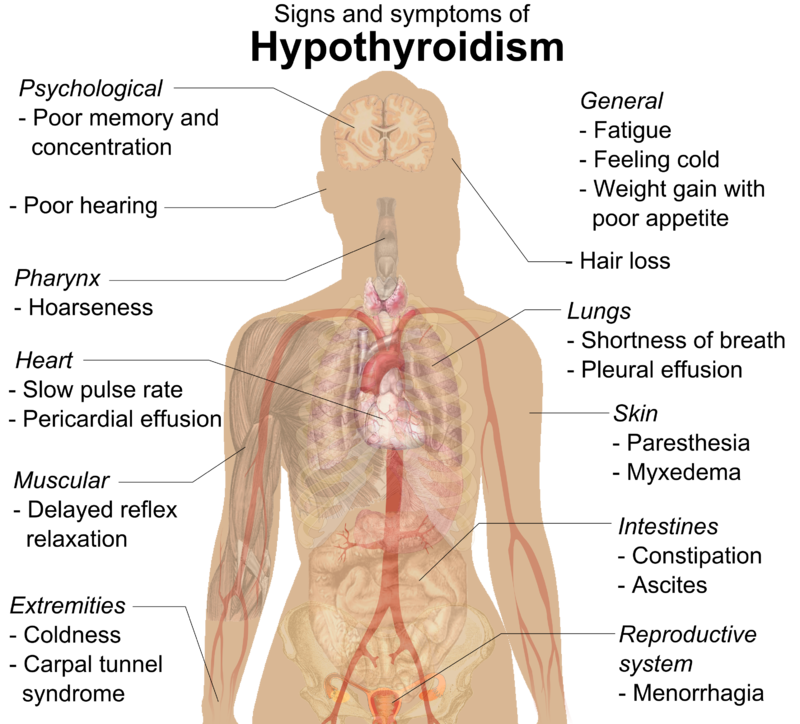
Symptoms of an underactive thyroid are often the same for pregnant women as for other people with hypothyroidism. Symptoms include
- extreme tiredness
- trouble dealing with cold
- muscle cramps
- severe constipation
- problems with memory or concentration
Most cases of hypothyroidism in pregnancy are mild and may not have symptoms.
What causes hypothyroidism in pregnancy?
Hypothyroidism in pregnancy is usually caused by Hashimoto’s disease and occurs in 2 to 3 out of every 100 pregnancies.1 Hashimoto’s disease is an autoimmune disorder. In Hashimoto’s disease, the immune system makes antibodies that attack the thyroid, causing inflammation and damage that make it less able to make thyroid hormones.
How can hypothyroidism affect me and my baby?
Untreated hypothyroidism during pregnancy can lead to
- preeclampsia—a dangerous rise in blood pressure in late pregnancy
- anemia
- miscarriage
- low birthweight
- stillbirth
- congestive heart failure, rarely
These problems occur most often with severe hypothyroidism.
Because thyroid hormones are so important to your baby’s brain and nervous system development, untreated hypothyroidism—especially during the first trimester—can cause low IQ and problems with normal development.
How do doctors diagnose hypothyroidism in pregnancy?
Your doctor will review your symptoms and do some blood tests to measure your thyroid hormone levels. Your doctor may also look for certain antibodies in your blood to see if Hashimoto’s disease is causing your hypothyroidism. Learn more about thyroid tests and what the results mean.
How do doctors treat hypothyroidism during pregnancy?
Treatment for hypothyroidism involves replacing the hormone that your own thyroid can no longer make. Your doctor will most likely prescribe levothyroxine, a thyroid hormone medicine that is the same as T4, one of the hormones the thyroid normally makes. Levothyroxine is safe for your baby and especially important until your baby can make his or her own thyroid hormone.
Your thyroid makes a second type of hormone, T3. Early in pregnancy, T3 can’t enter your baby’s brain like T4 can. Instead, any T3 that your baby’s brain needs is made from T4. T3 is included in a lot of thyroid medicines made with animal thyroid, such as Armour Thyroid, but is not useful for your baby’s brain development. These medicines contain too much T3 and not enough T4, and should not be used during pregnancy. Experts recommend only using levothyroxine (T4) while you’re pregnant.
These medicines contain too much T3 and not enough T4, and should not be used during pregnancy. Experts recommend only using levothyroxine (T4) while you’re pregnant.
Some women with subclinical hypothyroidism—a mild form of the disease with no clear symptoms—may not need treatment.
Your doctor may prescribe levothyroxine to treat your hypothyroidism.If you had hypothyroidism before you became pregnant and are taking levothyroxine, you will probably need to increase your dose. Most thyroid specialists recommend taking two extra doses of thyroid medicine per week, starting right away. Contact your doctor as soon as you know you’re pregnant.
Your doctor will most likely test your thyroid hormone levels every 4 to 6 weeks for the first half of your pregnancy, and at least once after 30 weeks.1 You may need to adjust your dose a few times.
Postpartum Thyroiditis
What is postpartum thyroiditis?
Postpartum thyroiditis is an inflammation of the thyroid that affects about 1 in 20 women during the first year after giving birth1 and is more common in women with type 1 diabetes.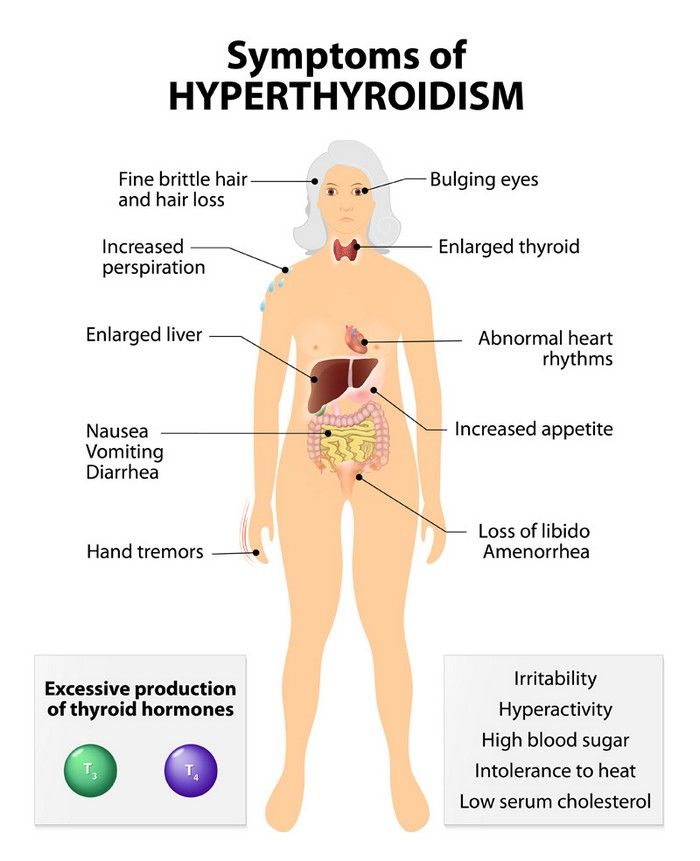 The inflammation causes stored thyroid hormone to leak out of your thyroid gland. At first, the leakage raises the hormone levels in your blood, leading to hyperthyroidism. The hyperthyroidism may last up to 3 months. After that, some damage to your thyroid may cause it to become underactive. Your hypothyroidism may last up to a year after your baby is born. However, in some women, hypothyroidism doesn’t go away.
The inflammation causes stored thyroid hormone to leak out of your thyroid gland. At first, the leakage raises the hormone levels in your blood, leading to hyperthyroidism. The hyperthyroidism may last up to 3 months. After that, some damage to your thyroid may cause it to become underactive. Your hypothyroidism may last up to a year after your baby is born. However, in some women, hypothyroidism doesn’t go away.
Not all women who have postpartum thyroiditis go through both phases. Some only go through the hyperthyroid phase, and some only the hypothyroid phase.
What are the symptoms of postpartum thyroiditis?
The hyperthyroid phase often has no symptoms—or only mild ones. Symptoms may include irritability, trouble dealing with heat, tiredness, trouble sleeping, and fast heartbeat.
Symptoms of the hypothyroid phase may be mistaken for the “baby blues”—the tiredness and moodiness that sometimes occur after the baby is born. Symptoms of hypothyroidism may also include trouble dealing with cold; dry skin; trouble concentrating; and tingling in your hands, arms, feet, or legs.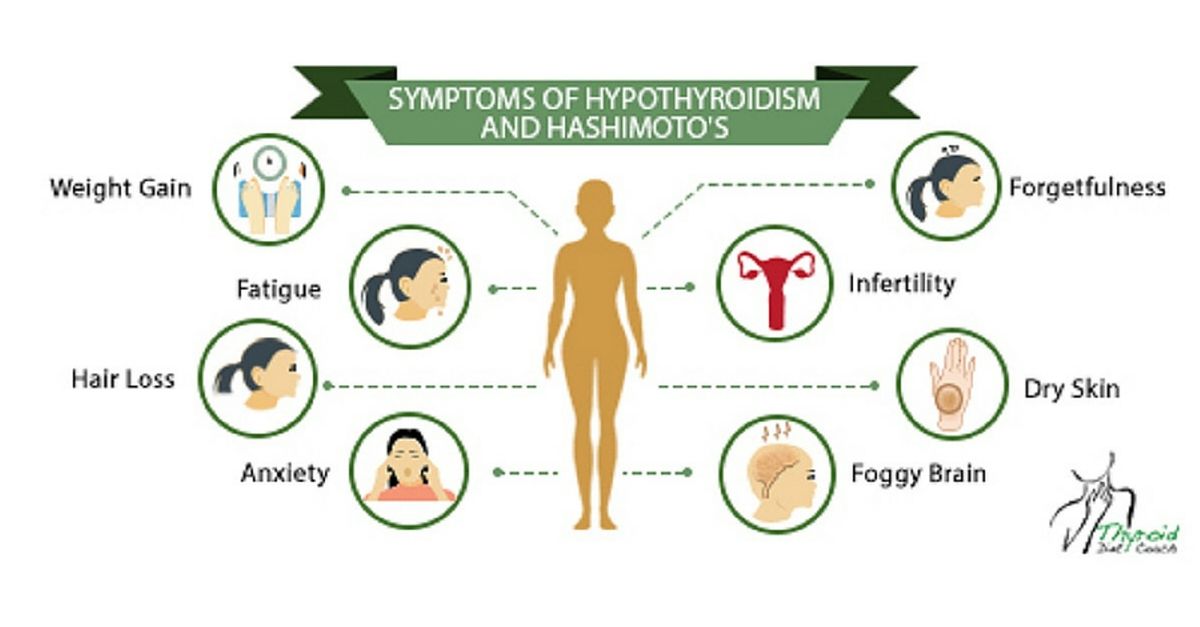 If these symptoms occur in the first few months after your baby is born or you develop postpartum depression, talk with your doctor as soon as possible.
If these symptoms occur in the first few months after your baby is born or you develop postpartum depression, talk with your doctor as soon as possible.
What causes postpartum thyroiditis?
Postpartum thyroiditis is an autoimmune condition similar to Hashimoto’s disease. If you have postpartum thyroiditis, you may have already had a mild form of autoimmune thyroiditis that flares up after you give birth.
Postpartum thyroiditis may last up to a year after your baby is born.How do doctors diagnose postpartum thyroiditis?
If you have symptoms of postpartum thyroiditis, your doctor will order blood tests to check your thyroid hormone levels.
How do doctors treat postpartum thyroiditis?
The hyperthyroid stage of postpartum thyroiditis rarely needs treatment. If your symptoms are bothering you, your doctor may prescribe a beta-blocker, a medicine that slows your heart rate. Antithyroid medicines are not useful in postpartum thyroiditis, but if you have Grave’s disease, it may worsen after your baby is born and you may need antithyroid medicines.
You’re more likely to have symptoms during the hypothyroid stage. Your doctor may prescribe thyroid hormone medicine to help with your symptoms. If your hypothyroidism doesn’t go away, you will need to take thyroid hormone medicine for the rest of your life.
Is it safe to breastfeed while I’m taking beta-blockers, thyroid hormone, or antithyroid medicines?
Certain beta-blockers are safe to use while you’re breastfeeding because only a small amount shows up in breast milk. The lowest possible dose to relieve your symptoms is best. Only a small amount of thyroid hormone medicine reaches your baby through breast milk, so it’s safe to take while you’re breastfeeding. However, in the case of antithyroid drugs, your doctor will most likely limit your dose to no more than 20 milligrams (mg) of methimazole or, less commonly, 400 mg of PTU.
Thyroid Disease and Eating During Pregnancy
What should I eat during pregnancy to help keep my thyroid and my baby’s thyroid working well?
Because the thyroid uses iodine to make thyroid hormone, iodine is an important mineral for you while you’re pregnant. During pregnancy, your baby gets iodine from your diet. You’ll need more iodine when you’re pregnant—about 250 micrograms a day.1 Good sources of iodine are dairy foods, seafood, eggs, meat, poultry, and iodized salt—salt with added iodine. Experts recommend taking a prenatal vitamin with 150 micrograms of iodine to make sure you’re getting enough, especially if you don’t use iodized salt.1 You also need more iodine while you’re breastfeeding since your baby gets iodine from breast milk. However, too much iodine from supplements such as seaweed can cause thyroid problems. Talk with your doctor about an eating plan that’s right for you and what supplements you should take. Learn more about a healthy diet and nutrition during pregnancy.
During pregnancy, your baby gets iodine from your diet. You’ll need more iodine when you’re pregnant—about 250 micrograms a day.1 Good sources of iodine are dairy foods, seafood, eggs, meat, poultry, and iodized salt—salt with added iodine. Experts recommend taking a prenatal vitamin with 150 micrograms of iodine to make sure you’re getting enough, especially if you don’t use iodized salt.1 You also need more iodine while you’re breastfeeding since your baby gets iodine from breast milk. However, too much iodine from supplements such as seaweed can cause thyroid problems. Talk with your doctor about an eating plan that’s right for you and what supplements you should take. Learn more about a healthy diet and nutrition during pregnancy.
Clinical Trials
The National Institute of Diabetes and Digestive and Kidney Diseases (NIDDK) and other components of the National Institutes of Health (NIH) conduct and support research into many diseases and conditions.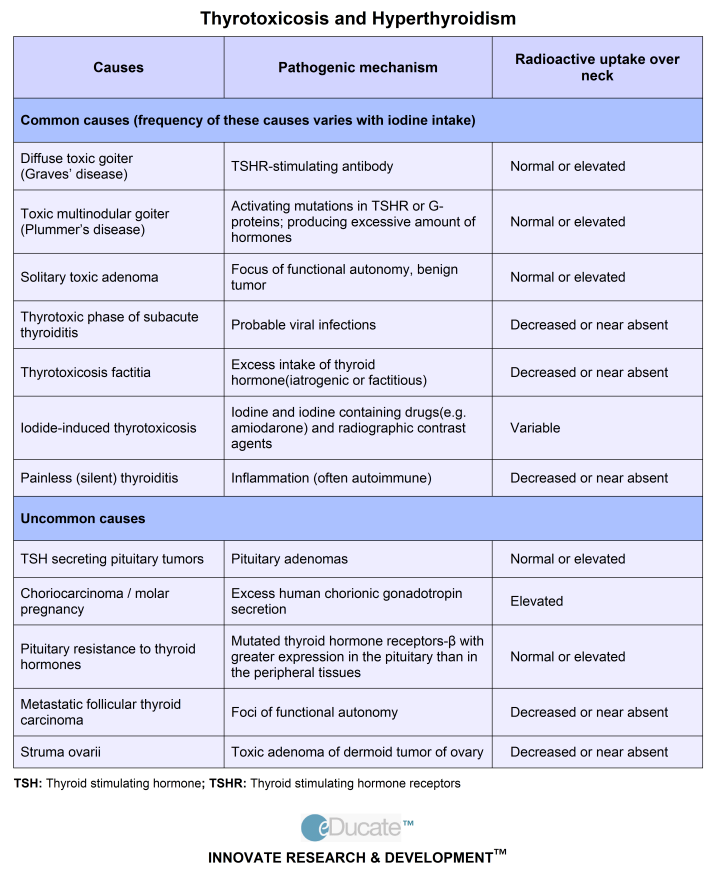
What are clinical trials, and are they right for you?
Clinical trials are part of clinical research and at the heart of all medical advances. Clinical trials look at new ways to prevent, detect, or treat disease. Researchers also use clinical trials to look at other aspects of care, such as improving the quality of life for people with chronic illnesses. Find out if clinical trials are right for you.
What clinical trials are open?
Clinical trials that are currently open and are recruiting can be viewed at www.ClinicalTrials.gov.
References
Hyperthyroidism during pregnancy - causes, symptoms, diagnosis and treatment
Hyperthyroidism during pregnancy is an increase in the content of thyroid hormones that occurs before conception or is associated with gestation. Manifested by weakness, fatigue, sweating, nausea, vomiting, increased appetite, increased defecation, irritability, insomnia, hand tremor, palpitations, in some patients - an increase in the thyroid gland, exophthalmos, infiltrative dermatopathy of the lower extremities.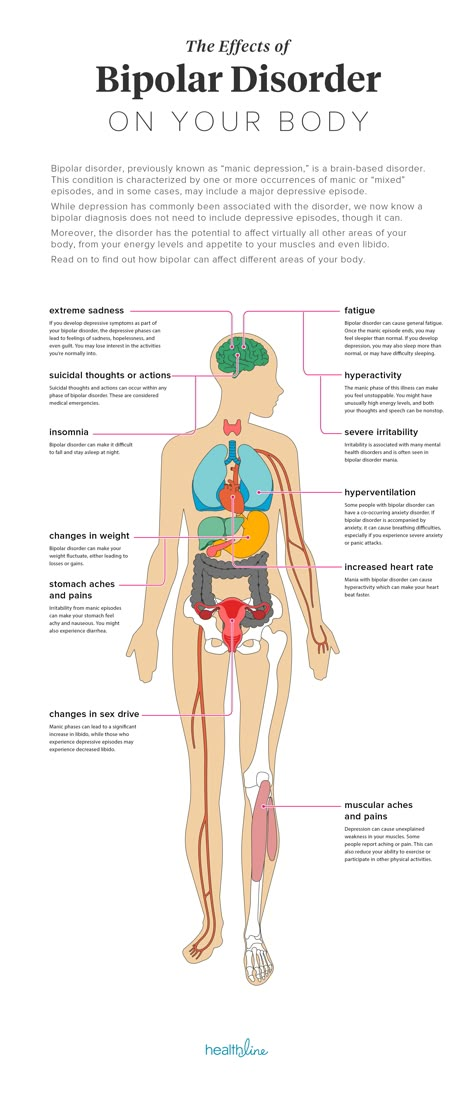 It is diagnosed on the basis of test data on the content of T3, T4, TSH. For treatment, thyreostatics, β-blockers are used, with a resistant course, bilateral subtotal resection of the thyroid gland is performed.
It is diagnosed on the basis of test data on the content of T3, T4, TSH. For treatment, thyreostatics, β-blockers are used, with a resistant course, bilateral subtotal resection of the thyroid gland is performed.
General information
According to endocrinology studies, the prevalence of hyperthyroidism reaches 1%, in more than half of the cases the disorder is subclinical. The frequency of thyrotoxicosis during gestation is 0.05-0.4%. In 85-90%, the disorder occurs before the onset of pregnancy and is due to Graves' disease. However, in some women, hyperthyroidism is gestational, formed either in the first trimester as a result of compensatory hormonal changes in the body, or after childbirth due to the development of autoimmune processes. The relevance of timely detection of pathology is associated with a high risk of complications in severe thyrotoxicosis.
Hyperthyroidism during pregnancy
Causes
There are several groups of pathological conditions that are manifested by increased synthesis and release of thyroid hormones in pregnant women. Only in 8-10% of patients, hyperthyroidism is associated with physiological processes occurring during gestation and after childbirth. In other cases, the level of thyroxine and triiodothyronine increases due to diseases that arose before pregnancy. The main etiological factors are:
Only in 8-10% of patients, hyperthyroidism is associated with physiological processes occurring during gestation and after childbirth. In other cases, the level of thyroxine and triiodothyronine increases due to diseases that arose before pregnancy. The main etiological factors are:
- Hyperstimulation of thyrotropin receptors . The thyroid gland produces an excess amount of hormones in thyrotropinomas - pituitary adenomas that secrete TSH, the stimulating effect of autoimmune complexes in Graves' disease, increased sensitivity of thyrotropic receptors due to mutations in toxic multinodular goiter or a solitary node. At 8-14 weeks of pregnancy, the synthesis of hormones potentiates chorionic gonadotropin, similar in structure to thyrotropin.
- Thyroid destruction . With subacute granulomatous thyroiditis, Hashimoto's thyroiditis, postpartum autoimmune thyroiditis, which occurs in some women in the first months after childbirth, the thyroid tissue is destroyed, and the hormones contained in it enter the bloodstream.
 At the initial stages of inflammation, hyperthyroidism develops, which can later be replaced by hypothyroidism. Similar processes are possible when prescribing α-interferon, amiodarone, and in some cases lithium preparations.
At the initial stages of inflammation, hyperthyroidism develops, which can later be replaced by hypothyroidism. Similar processes are possible when prescribing α-interferon, amiodarone, and in some cases lithium preparations. - High blood iodine level . Sometimes hyperthyroidism is observed against the background of increased activity of functionally autonomous areas of the thyroid gland, which use excess iodine to produce hormones. Usually the condition is transient and normalizes after the trace element is removed from the body. Its causes may be the intake of iodine preparations (cordarone, expectorants), radiation studies with iodine-containing contrast, less often - the use of foods rich in iodine.
Extremely rarely, hyperthyroidism during pregnancy is provoked by pulmonary metastases of follicular thyroid cancer and ovarian teratomas containing sufficient thyroid tissue. The occurrence of transient drug-induced thyrotoxicosis is possible after an accidental or intentional intake of thyroid hormones in an increased dosage.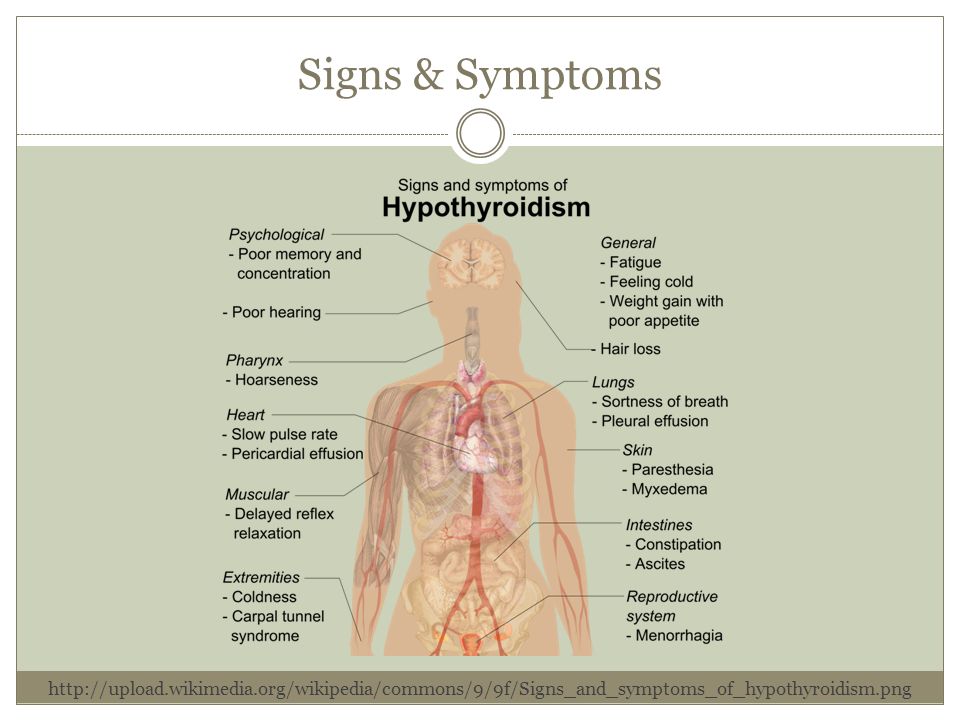
Pathogenesis
The key moment in the development of hyperthyroidism is an increase in the secreting function of TSH-sensitive or functionally autonomous thyroid cells or the release of hormones from the destroyed thyroid tissue. The occurrence or intensification of these processes is facilitated by changes that occur during gestation. At the beginning of pregnancy, the activity of the gland increases physiologically, which is associated with the importance of thyroid hormones for the normal functioning of the corpus luteum, which maintains pregnancy in the early stages. Estrogens, the level of which gradually increases, potentiate the production of thyroxine-binding globulin (TSG) in the liver.
Compensatory enhancement of thyrotropin synthesis contributes to maintaining the normal level of biologically active free fractions of thyroid hormones with an increase in their total content. Since the fetal thyroid gland begins to secrete hormones only from the 12th week of pregnancy, in the 1st trimester the child's needs for triiodothyronine and thyroxine are satisfied due to their temporary overproduction in the pregnant woman's body under the influence of hCG.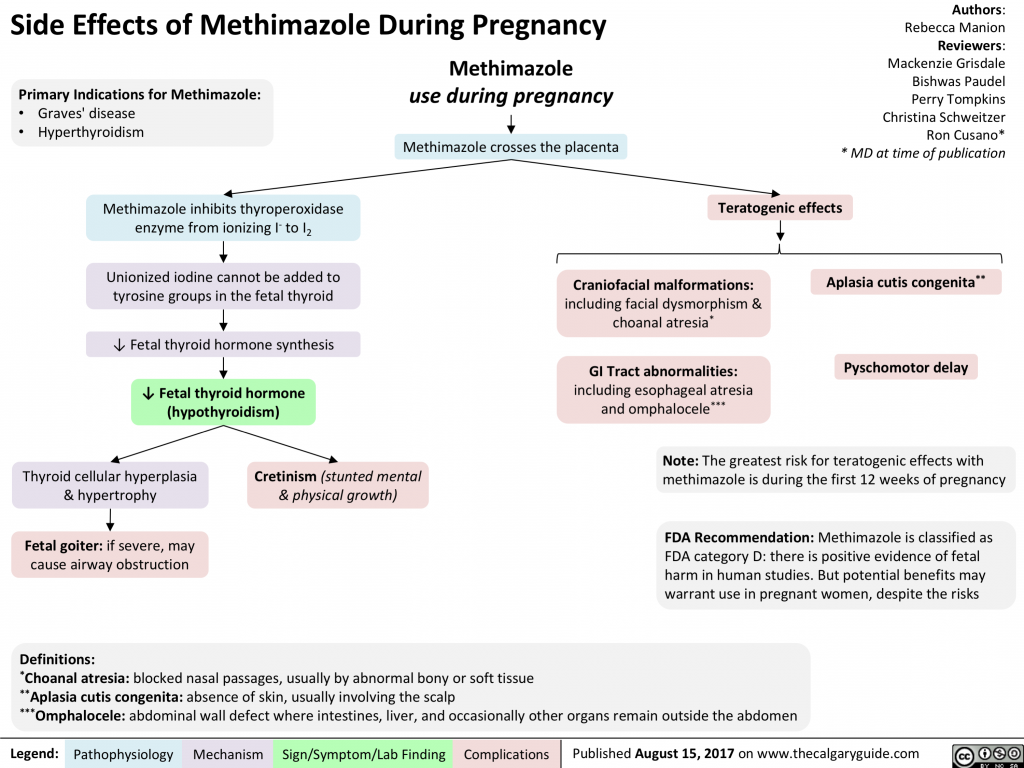 Immune restructuring after childbirth may be accompanied by the formation of antibodies to thyroid tissue and the development of transient inflammation with signs of hyperthyroidism, which is often followed by a decrease in thyroid function.
Immune restructuring after childbirth may be accompanied by the formation of antibodies to thyroid tissue and the development of transient inflammation with signs of hyperthyroidism, which is often followed by a decrease in thyroid function.
Classification
Hyperthyroidism during pregnancy is systematized taking into account the same criteria as outside the gestational period - the etiological factor and the severity of clinical manifestations. For reasons, there are primary thyrotoxicosis, caused by hyperproduction of thyroid hormones by the thyroid gland, secondary, arising against the background of pituitary disorders, and tertiary, provoked by hypothalamic dysfunction. Separately, there are variants of the disorder that are caused by the secretory activity of the thyroid tissue outside the thyroid gland, associated with its destruction or an overdose of hormonal drugs. Depending on the severity, the following variants of hyperthyroidism are distinguished:
- Subclinical .
 Runs asymptomatic. With a slightly reduced level of thyrotropin, a normal content of thyroxine is noted. The risk of obstetric and extragenital complications is minimal.
Runs asymptomatic. With a slightly reduced level of thyrotropin, a normal content of thyroxine is noted. The risk of obstetric and extragenital complications is minimal. - Manifest . There is a characteristic clinical picture. A significant decrease in the concentration of thyroid-stimulating hormone and an increase in the level of thyroxin are revealed. The risk of pregnancy complications increases.
- Complicated . Thyroid hormones are toxic to the body. Atrial fibrillation, dystrophy of parenchymal organs, adrenal, heart failure, and other life-threatening disorders develop.
Symptoms of hyperthyroidism during pregnancy
The clinical symptoms of thyrotoxicosis usually do not depend on the causes that caused it. With a latent course of hyperthyroidism, symptoms can be determined that indicate an acceleration of metabolism - insufficient weight gain, warm skin, increased sweating, fatigue, muscle weakness. With a manifest course, pregnant women complain of heat intolerance, nausea, vomiting, increased appetite, increased urge to defecate, insomnia, trembling fingers, palpitations in the precordial region, neck, head, and abdominal cavity. Some patients have subfebrile condition.
With a manifest course, pregnant women complain of heat intolerance, nausea, vomiting, increased appetite, increased urge to defecate, insomnia, trembling fingers, palpitations in the precordial region, neck, head, and abdominal cavity. Some patients have subfebrile condition.
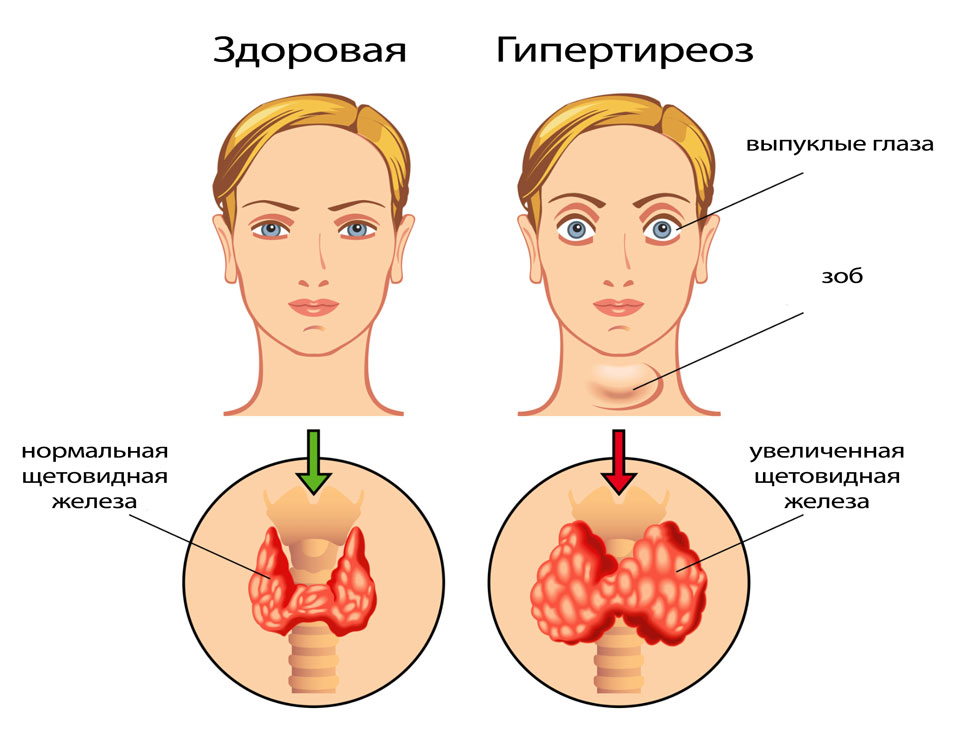
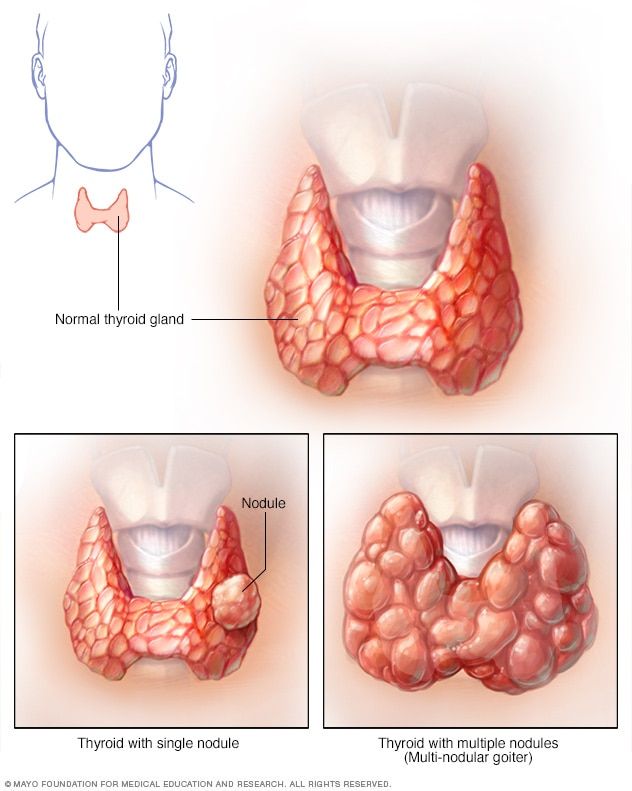
The woman looks fussy, touchy, irritable, tearful. With diffuse hyperplasia of the thyroid tissue, a thickening of the lower part of the neck becomes noticeable. Sometimes nodular formations are determined in the thyroid gland. The characteristic signs of hyperthyroidism in Graves' disease are infiltrative ophthalmopathy and dermatopathy. In 60% of pregnant women with toxic goiter, there are pains in the orbits, lacrimation, redness of the conjunctiva, sclera, photophobia, bulging eyes (exophthalmos), double vision when viewing objects. Thyroid dermatopathy is manifested by itching, redness of the anterior surface of the legs, the formation of nodes, extensive non-inflammatory infiltrates. The skin of the toes is less commonly affected.
The skin of the toes is less commonly affected.
Complications
With transient hyperthyroidism occurring in the first trimester, early toxicosis with uncontrollable vomiting of pregnant women is more often detected. According to the observations of specialists in the field of obstetrics, a complicated course of gestation is usually found in patients who suffer from toxic goiter. A significant increase in the concentration of thyroid hormones affects the processes of implantation and disrupts embryogenesis, which leads to spontaneous miscarriage. With thyrotoxicosis, the risk of premature birth, stillbirth, gestosis with severe hypertensive syndrome, placental abruption, and coagulopathic bleeding increases. The course of the disease can be complicated by thyrotoxic crisis, heart failure.
Against the background of cardiovascular disorders characteristic of hyperthyroidism, fetoplacental insufficiency is more often formed, leading to a delay in fetal development. The toxic effect of thyroid hormones increases the likelihood of anatomical developmental anomalies, including those incompatible with life. In 2-3% of pregnant women with hyperthyroidism, the transplacental transfer of autoantibodies to thyrotropic receptors contributes to the occurrence of intrauterine and neonatal thyrotoxicosis with fetal hypotrophy, increased neuromuscular excitability, and impaired psychomotor development of the newborn.
The toxic effect of thyroid hormones increases the likelihood of anatomical developmental anomalies, including those incompatible with life. In 2-3% of pregnant women with hyperthyroidism, the transplacental transfer of autoantibodies to thyrotropic receptors contributes to the occurrence of intrauterine and neonatal thyrotoxicosis with fetal hypotrophy, increased neuromuscular excitability, and impaired psychomotor development of the newborn.
Diagnosis
In the presence of anamnestic information about thyroid diseases with an increase in its secretory activity, the diagnosis is not difficult. If you suspect the development or manifestation of thyrotoxicosis during the gestation period, tests are prescribed to detect hormonal imbalance. Laboratory markers of hyperthyroidism during pregnancy are:
- The content of thyroid hormones . In patients with a subclinical course, the indicator may remain normal. With the manifestation of the disease, the concentration of triiodothyronine (T3) and thyroxine (T4), especially free forms, increases.
- Thyrotropin level . The content of TSH decreases both in subclinical and manifest primary hyperthyroidism. Such a violation is associated with the suppressive effect of thyroid hormones that circulate in the blood of a pregnant woman.
- Determination of AT rTTG . Specific immunoglobulins interact with thyroid tissue receptors, stimulating its secretory function. The detection of antibodies serves as a marker of autoimmune thyroid disease.
To clarify the causes of thyrotoxicosis, the level of thyroxin-binding globulin is additionally assessed, the test of quenched thyroid hormones is performed, ultrasound and dopplerography of the thyroid gland are performed. Radiation research methods during pregnancy are not recommended due to possible damaging effects on the fetus. An important task of the examination is the differential diagnosis between transient gestational hyperthyroidism and an increase in the level of thyroid hormones due to damage to the tissues of the thyroid gland or other causes. In addition to an obstetrician-gynecologist and an endocrinologist, a neurosurgeon, an oncologist, an immunologist, a toxicologist, a cardiologist, an ophthalmologist, and a dermatologist consult the patient according to indications.
In addition to an obstetrician-gynecologist and an endocrinologist, a neurosurgeon, an oncologist, an immunologist, a toxicologist, a cardiologist, an ophthalmologist, and a dermatologist consult the patient according to indications.
Treatment of hyperthyroidism in pregnancy
For women with transient subclinical thyrotoxicosis, dynamic monitoring with regular laboratory control is recommended. The appointment of active methods of treatment is justified in case of manifest and complicated course of the disease. With therapeutically resistant hyperthyroidism, an artificial termination of pregnancy is carried out for medical reasons for up to 12 weeks. The choice of drugs for the treatment of thyrotoxicosis is determined by the causes that caused the disorder. The main difficulty of therapy is the impossibility of using drugs containing radioactive iodine during pregnancy. With diffuse toxic goiter, which is most often detected with an increased content of T3 and T4 in pregnant women, the following is prescribed:
- Antithyroid drugs .
 By blocking thyroid peroxidase, thyreostatics prevent the organization of iodides and the condensation of iodotyrosines, and inhibit the peripheral conversion of thyroxine to triiodothyronine. They allow to quickly improve the condition in 20-50% of patients.
By blocking thyroid peroxidase, thyreostatics prevent the organization of iodides and the condensation of iodotyrosines, and inhibit the peripheral conversion of thyroxine to triiodothyronine. They allow to quickly improve the condition in 20-50% of patients. - β-blockers . Indicated to eliminate the effects of adrenergic stimulation that has arisen against the background of hyperthyroidism. Effectively reduce hand tremor, tachycardia, rhythm disturbances, emotional disorders, heat intolerance, loose stools, proximal myopathy.
- Iodine preparations . During pregnancy, they are rarely used due to possible toxic effects (conjunctivitis, rash, inflammation of the salivary glands). They make it possible to quickly inhibit the release of thyroid hormones and the organization of iodine. Recommended for thyrotoxic crises and for preoperative preparation.
Usually, drug therapy is carried out in the 1st trimester, when the course of Graves' disease is aggravated by physiological changes during pregnancy. Patients with severe hyperthyroidism, intolerance to thyreostatics, ineffectiveness of conservative treatment, compression of adjacent organs by goiter, and suspected thyroid cancer require surgery. The intervention is performed in the 2nd trimester, when the risk of spontaneous abortion is minimal. The volume of resection is determined by the severity of the disorder. As a rule, bilateral subtotal strumectomy is performed.
Patients with severe hyperthyroidism, intolerance to thyreostatics, ineffectiveness of conservative treatment, compression of adjacent organs by goiter, and suspected thyroid cancer require surgery. The intervention is performed in the 2nd trimester, when the risk of spontaneous abortion is minimal. The volume of resection is determined by the severity of the disorder. As a rule, bilateral subtotal strumectomy is performed.
Euthyroid vaginal delivery with adequate analgesia, fetal and hemodynamic monitoring is the preferred method of delivery. Usually, the birth process proceeds quickly, its duration in primiparous does not exceed 10 hours. Caesarean section is carried out in the presence of obstetric indications (wrong position of the fetus, narrow pelvis, cord entanglement, placenta previa, etc.). With exacerbation of hyperthyroidism in the postpartum period, suppression of lactation and the appointment of thyreostatic drugs are recommended.
Prognosis and prevention
Timely diagnosis of hyperthyroidism and selection of an adequate treatment regimen allows most patients to endure pregnancy normally. From 24-28 weeks, the severity of the disorder decreases, spontaneous remission of the thyroid disease is possible. Pregnancy in women with pathologies accompanied by thyrotoxicosis should be planned taking into account the recommendations of the endocrinologist. The optimal time for conception is a period of stable remission with euthyroidism 3 or more months after the end of drug treatment. For preventive purposes, early registration in the antenatal clinic is indicated.
From 24-28 weeks, the severity of the disorder decreases, spontaneous remission of the thyroid disease is possible. Pregnancy in women with pathologies accompanied by thyrotoxicosis should be planned taking into account the recommendations of the endocrinologist. The optimal time for conception is a period of stable remission with euthyroidism 3 or more months after the end of drug treatment. For preventive purposes, early registration in the antenatal clinic is indicated.
Thyroid disorders and pregnancy | Burumkulova
In their practice, both endocrinologists and obstetrician-gynecologists often encounter various diseases of the thyroid gland (thyroid gland) in pregnant women, which is of significant clinical and scientific interest both for studying the pathology of these disorders and in terms of their treatment.
As you know, pregnancy often leads to goiter. An increase in the size and volume of the thyroid gland during pregnancy is observed due to both a more intensive blood supply to the thyroid tissue and an increase in the mass of the thyroid tissue.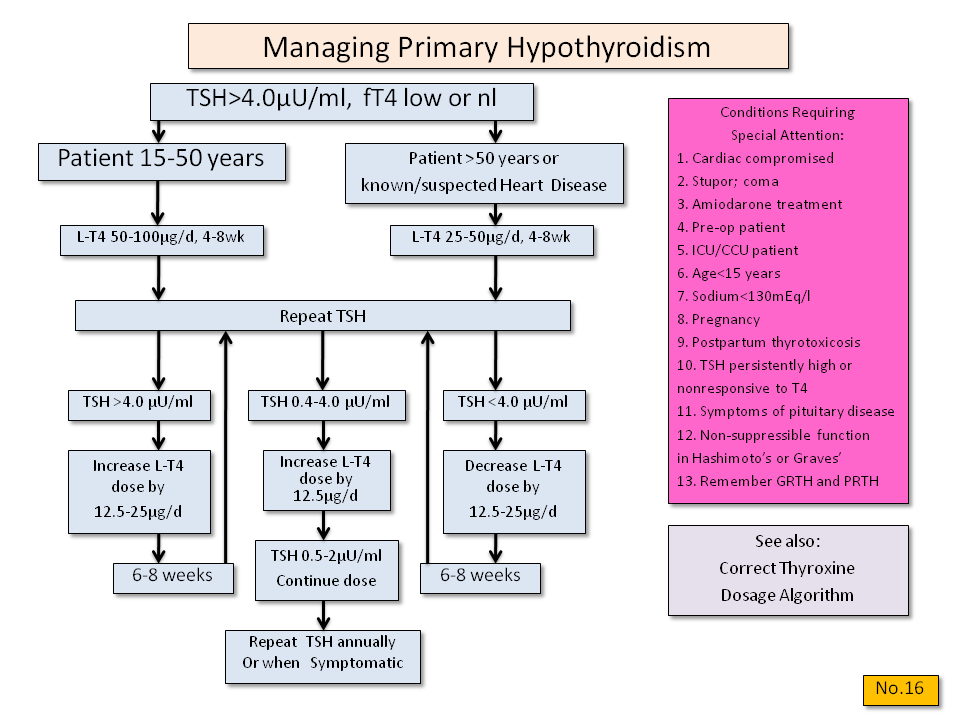 3 factors can stimulate thyroid function during pregnancy: an increase in the degree of binding of thyroid hormones (TG) to blood proteins, an increase in the level of chorionic gonadotropin (CG) in the blood of pregnant women, and an insufficient supply of iodine to the thyroid gland due to increased excretion of iodine in the urine during pregnancy (see . picture).
3 factors can stimulate thyroid function during pregnancy: an increase in the degree of binding of thyroid hormones (TG) to blood proteins, an increase in the level of chorionic gonadotropin (CG) in the blood of pregnant women, and an insufficient supply of iodine to the thyroid gland due to increased excretion of iodine in the urine during pregnancy (see . picture).
Increased binding of TG to blood proteins. More than 99% of TG circulating in the blood is bound to plasma proteins: thyroxine-binding globulin (TSG), thyroxine-binding prealbumin and albumin. The relative distribution of the amount of TG binding to various binding proteins directly depends on the degree of their affinity and concentration. 80% of TG is associated with TSH. The bound and inactive TG fractions are in equilibrium with the "free" unbound fraction, which represents only a small fraction of all circulating TG: 0.03-0.04% for free thyroxine (swT 4 ) and 0.3-0.5% for free triiodothyronine (swt 3 ).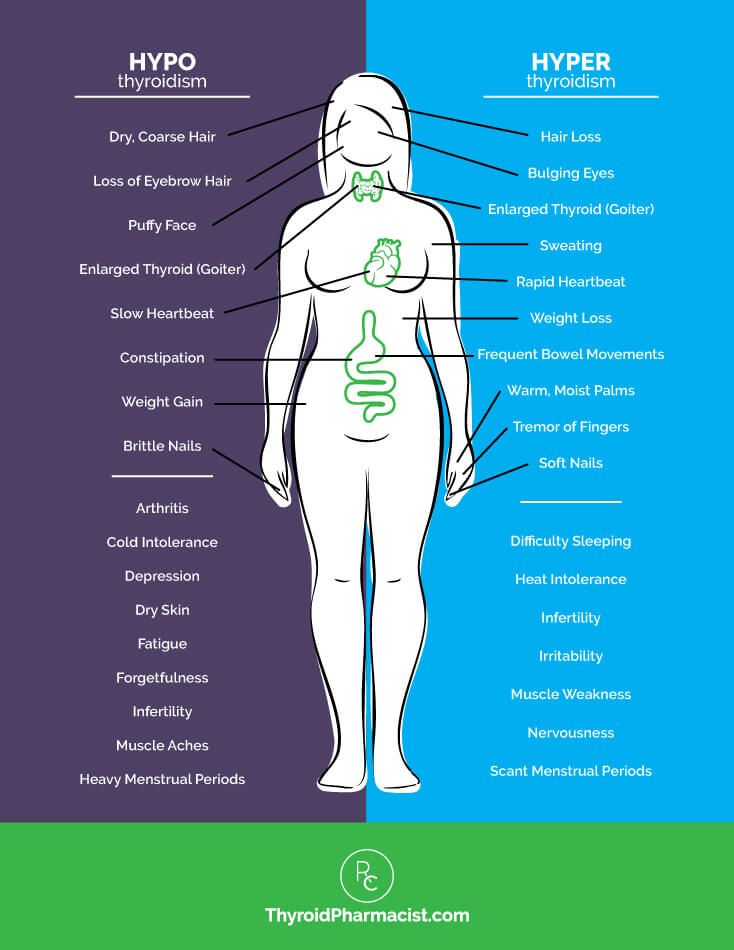 However, it is this fraction that provides all the metabolic and biological activity of TH.
However, it is this fraction that provides all the metabolic and biological activity of TH.
During pregnancy, already a few weeks after conception, the serum level of TSH progressively increases as a result of stimulation by a significant amount of estrogens produced by the placenta. Then the level of TSH reaches a plateau, which is maintained until the moment of delivery. Conversely, the level of 2 other circulating binding proteins tends to decrease, mainly as a result of passive
Scheme of thyroid stimulation during pregnancy
dilution due to increased vascular pool (blood depot).
Increased TSH production during pregnancy results in an increase in total TG levels. The levels of total T 4 (vT 4 ) and T 3 (vT 3 ) increase significantly during the first half of pregnancy and reach a plateau by the 20th week, remaining at the same level thereafter. Transient decrease in the amount of svt 4 and svTz on the feedback principle stimulates the release of thyroid-stimulating hormone (TSH) and restoration of homeostasis of the level of free forms of TG.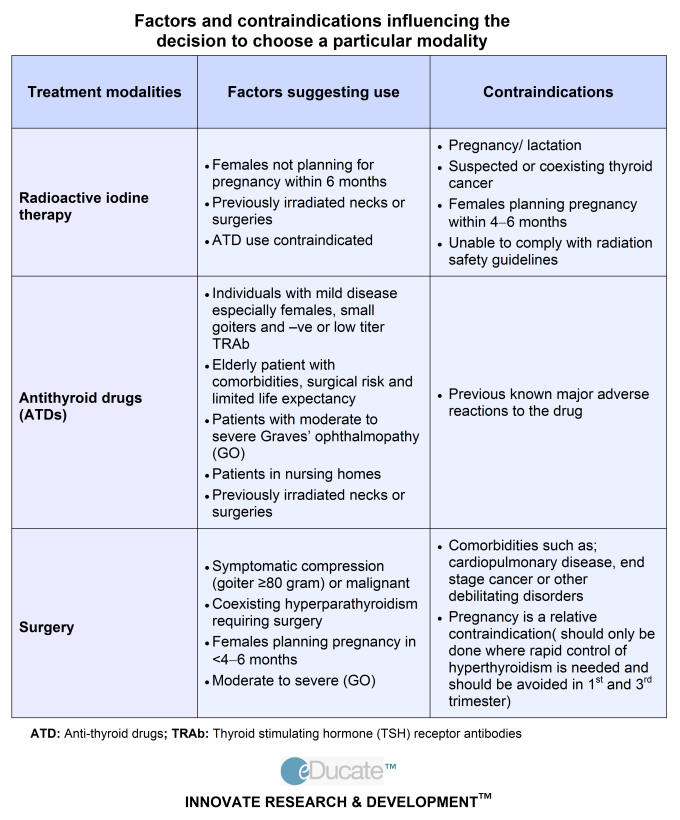
Adequate maintenance of thyroid homeostasis is disturbed in about 1/3 of pregnant women, which leads to the development of a state of relative hypothyroxinemia.
Stimulation of thyroid function during pregnancy hCG. CG is secreted by the placenta only in primates. It is produced in large quantities by placental syncytiotrophoblasts, especially in the first quarter of pregnancy. The most important function of hCG is the stimulation of steroidogenesis, first in the corpus luteum, then in the placenta.
The value of hCG for thyroid stimulation in women during pregnancy is not fully understood. It is known that there is a correlation between the suppression of TSH secretion and an increase in the concentration of hCG, as well as between the level of hCG and the level of fT 4 . CG is able to have a direct stimulating effect on the mother's thyroid gland (and this effect is most pronounced at the end of the first trimester of pregnancy) due to the molecular similarity of CG to TSH. Acting in early pregnancy as a weak 'analogue' of TSH, hCG is responsible for the slight increase in serum FTT levels 4 and svt 3 and, as a result, for a decrease in serum TSH levels. In the vast majority of healthy pregnant women, the stimulatory effect of CG on the thyroid gland is short and insignificant. However, in 1-2% of all pregnant women during the first trimester of pregnancy, there is a decrease in the concentration of TSH and an increase in the level of sT 3 , which is accompanied by a clinic of thyrotoxicosis. This syndrome is called ''gestational transient thyrotoxicosis'' (GTT).
Acting in early pregnancy as a weak 'analogue' of TSH, hCG is responsible for the slight increase in serum FTT levels 4 and svt 3 and, as a result, for a decrease in serum TSH levels. In the vast majority of healthy pregnant women, the stimulatory effect of CG on the thyroid gland is short and insignificant. However, in 1-2% of all pregnant women during the first trimester of pregnancy, there is a decrease in the concentration of TSH and an increase in the level of sT 3 , which is accompanied by a clinic of thyrotoxicosis. This syndrome is called ''gestational transient thyrotoxicosis'' (GTT).
Possible reasons for the increase in the level of CG and the development of GTT may be the following: 1) unbalanced production of CG due to transient overexpression of the gene encoding the P-subunit of CG; 2) changes in the degree of glycosylation of the CG molecule, which in turn leads to a prolongation of its half-life; 3) an increase in the mass of placental trophoblast syncytial cells in some women (for example, in multiple pregnancies). In multiple pregnancy, the concentration of hCG increases in proportion to the number of placentas.
In multiple pregnancy, the concentration of hCG increases in proportion to the number of placentas.
GTT is often accompanied by uncontrollable vomiting of pregnant women (hyperemesis gravidatum), which makes its diagnosis difficult due to the fact that nausea and vomiting are in principle characteristic of early pregnancy. This condition is usually transient and resolves by the second trimester of pregnancy. The diagnosis of HTT is made on the basis of an elevated level of hCG, a slightly suppressed concentration of TSH, an increase in serum levels of fT 4 and FT 3 to the levels characteristic of hyperthyroidism. Treatment with thyreostatics GTT is not indicated; with severe clinical symptoms, only a short course of β-blockers is sufficient.
Thus, it is important for clinicians to know that the symptoms of thyrotoxicosis during pregnancy have specific differences and may be the result of not only an autoimmune process in the thyroid gland, but also hormonal changes inherent in pregnancy itself.
Reduced availability of iodine while increasing the need for it during pregnancy. The increased need for iodine during pregnancy is due to two factors. On the one hand, during pregnancy, there is an additional loss of iodine from the mother's body due to increased renal clearance of iodide, on the other hand, the loss of iodide in the second half of pregnancy increases due to the fact that part of the maternal pool of inorganic iodide is consumed by the fetoplacental complex and is used for synthesis TG thyroid of the fetus.
For women living in countries with sufficient iodine intake (such as Japan, the United States or Scandinavia), iodine loss during pregnancy is not significant, since daily iodine intake is more than 150-200 mcg/day and remains satisfactory in throughout the entire pregnancy.
At the same time, in regions with moderate and severe iodine deficiency in the biosphere, which include the vast majority of Russia, reduced iodine intake (less than 100 µg/day) is a rather severe factor in thyroid stimulation during pregnancy.
The risk of developing thyroid disease during pregnancy is higher in women with a history of goiter (diffuse or nodular), and the number and size of the nodules may increase during pregnancy. Repeated pregnancy leads to a further increase in the size of the thyroid gland and increased nodulation.
In 1989, D. Glinoer et al. proposed a hypothesis according to which increased thyroid stimulation during pregnancy can lead to the formation of diffuse non-toxic goiter (DNG), and pregnancy is one of the factors causing pathological changes in the thyroid gland.
In clinical practice, the following biochemical parameters have been proposed to detect increased thyroid stimulation during pregnancy.
— Presence of relative hypothyroxinemia observed in about 1/3 of all pregnant women. For its diagnosis, certain ratios T 4 /TSG are recommended.
- Increased secretion of T 3 , manifested in an increase in the ratio of T 3 / T 4 more than 0. 025 and reflecting the stimulation of the thyroid gland in conditions of iodine deficiency.
025 and reflecting the stimulation of the thyroid gland in conditions of iodine deficiency.
- Change in the concentration of TSH in the blood. After the initial phase of suppression of the TSH level due to the high secretion of CG at the end of the first trimester of pregnancy, the TSH level progressively increases and its concentration by the time of delivery doubles in relation to the initial one. The increase in TSH levels usually remains within the normal range (<4 mU/l).
- Change in the concentration of thyroglobulin (Tg) in the blood serum. The serum level of Tg is a sensitive indicator of thyroid stimulation, which often increases during pregnancy: its increase is already observed in the first trimester, but is most pronounced in the third trimester and by the time of delivery. By the time of delivery, 60% of pregnant women have an elevated level of Tg in the blood.
An increase in Tg concentration correlates with other indicators of thyroid stimulation, such as a slight increase in TSH levels and an increase in the ratio of T 3 / T 4 more than 0.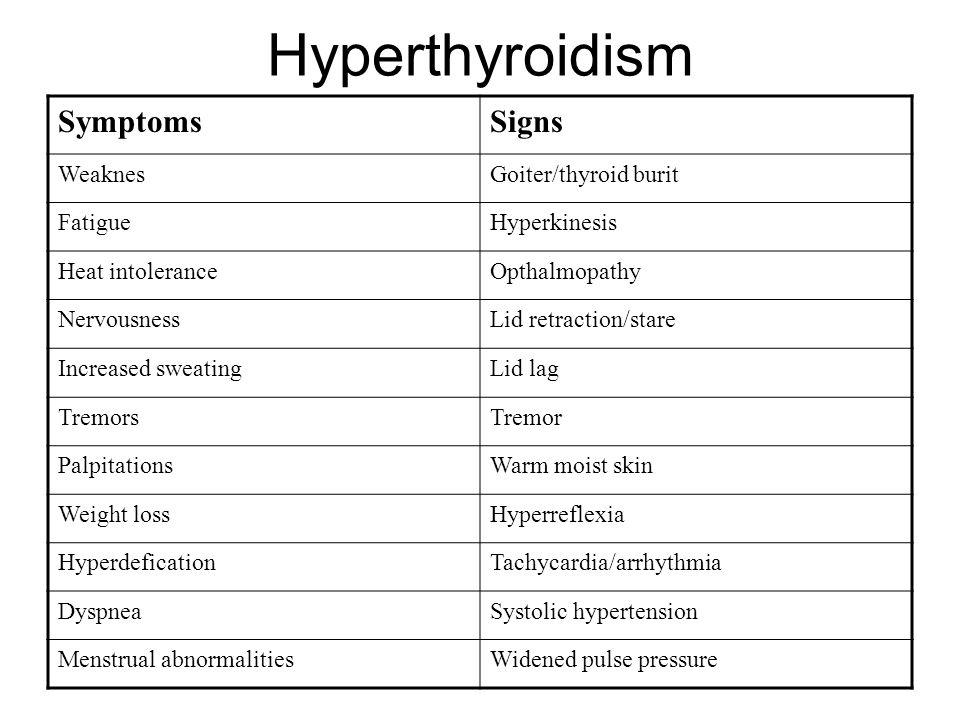 025. The presence of a correlation between the level of Tg and the volume of the thyroid gland (according to ultrasound - ultrasound confirms that the level of Tg in the blood is a fairly reliable biochemical marker of the goiterogenic effect of pregnancy.
025. The presence of a correlation between the level of Tg and the volume of the thyroid gland (according to ultrasound - ultrasound confirms that the level of Tg in the blood is a fairly reliable biochemical marker of the goiterogenic effect of pregnancy.
intellectual and physical development of the child.As is known, the thyroid gland of the fetus acquires the ability to concentrate iodine and synthesize iodothyronines at 10-12 weeks of intrauterine development.0123 4 , vT 4 and TSH reach adult levels around the 36th week of pregnancy.
The issue of placental permeability for triglycerides has been debatable for a long time. It is currently assumed that maternal and fetal thyroid glands are regulated autonomously, but not independently of each other. Apparently, the transplacental transfer of TG from the mother's body to the fetus is observed only at an early stage of intrauterine development.
In addition, the activity of the thyroid gland of the fetus is completely dependent on the intake of iodine from the mother's body. As a result of both insufficient intake of iodine in the mother’s body and a low intrathyroid iodine reserve, fetal thyroid stimulation occurs, which is reflected in a significant increase (compared with those of the mother) in the levels of neonatal TSH and Tg, as well as the development of goiter in the fetus. The development of hypothyroidism in the prenatal and neonatal periods can lead to an irreversible decrease in the child's mental development up to endemic cretinism.
As a result of both insufficient intake of iodine in the mother’s body and a low intrathyroid iodine reserve, fetal thyroid stimulation occurs, which is reflected in a significant increase (compared with those of the mother) in the levels of neonatal TSH and Tg, as well as the development of goiter in the fetus. The development of hypothyroidism in the prenatal and neonatal periods can lead to an irreversible decrease in the child's mental development up to endemic cretinism.
For the treatment of DND during pregnancy in regions with insufficient iodine intake, it is advisable to recommend iodine intake at the rate of 150-250 mcg/day. To do this, you can use the Antistrumine drug available in the pharmacy network (1000 μg of potassium iodide in 1 tablet), 1 tablet 1-2 times a week.
Another iodine preparation is "Potassium iodide-200" tablets, manufactured by Berlin-Chemie. They must be taken daily. An alternative may be imported multivitamins containing a daily dose of iodine (150 micrograms). As a rule, these prescriptions will be enough to prevent further growth of the goiter and even achieve a reduction in its volume.0005
As a rule, these prescriptions will be enough to prevent further growth of the goiter and even achieve a reduction in its volume.0005
In the presence of a large goiter before pregnancy or with its rapid growth at the beginning of pregnancy, a combination of iodine and thyroid hormones is justified: either Thyreocomb containing 70 µg T 4 , 10 µg T 3 and 150 µg iodine, or 50-100 mcg T 4 daily and additionally 1 tablet of antistrumine 2-3 times a week. This allows you to quickly and effectively restore the normal function of the mother's thyroid gland and prevent the goiter effect of pregnancy.
The development of hyperthyroidism during pregnancy is relatively rare and occurs in 0.05-3% of pregnant women. In most cases, its cause is diffuse toxic goiter (DTG), while toxic adenoma or multinodular toxic goiter are much less common.
The main difficulty in diagnosing thyrotoxicosis during pregnancy is that many clinical symptoms and signs of thyrotoxicosis can be masked by manifestations of a normal pregnancy (tachycardia, weakness, irritability, vegetative disorders, etc. ).
).
Diagnosis of DTG must be confirmed by history, ultrasound of the thyroid gland, as well as the study of the levels of TSH, sT 3 and especially sT 4 in the blood.
A typical error in the interpretation of the results of the study of hormonal function in pregnant women can be considered the determination of levels of vT 9vT 4 and vT 3 , which does not reflect the true functional state of the thyroid gland.
Thyrostatic drugs (mercasolil, methimazole, propylthiouracil) are preferred in all countries for the treatment of DTG in pregnant women. Surgical treatment is recommended only in exceptional cases, such as severe side effects, very large goiter, suspected malignancy, or the need to use high doses of thyreostatics to maintain maternal euthyroidism. The optimal time for subtotal resection of the thyroid gland is the second trimester of pregnancy. The appointment of iodides during pregnancy is contraindicated because of the risk of developing hypothyroidism in the fetus and goiter due to the Wolf-Chaikov effect.
What principles should be followed when treating a pregnant woman with DTG?
- The choice of a specific thyreostatic is determined both by the doctor's personal experience and the availability of a particular drug. In our country, mercasolil (1-methyl-2-mercaptoimidazole) or its analogues (methimazole, thiamazole) are more often used to treat DTG during pregnancy. Abroad, in a similar situation, preference is given to propylthiouracil (6-propyl-2-thiouracil). At present, a preparation of this group under the name Propicil (Kali-Khemi) has been registered and made available in Russia.
The frequency of side effects of therapy is the same for propylthiouracil and mercazolil. Both drugs cross the placenta, and excessive doses of them can cause the development of hypothyroidism and goiter in utero and neonatal periods.
Prescribing propylthiouracil during pregnancy, however, has a number of advantages. Firstly, the kinetics of propylthiouracil does not change during pregnancy, secondly, the half-life of propylthiouracil from the blood does not depend on the presence of hepatic or renal insufficiency, thirdly, propylthiouracil binds to proteins to a greater extent compared to mercazolil and has limited lipophilicity, which hinders its penetration through biological membranes such as the placenta and mammary gland epithelium.
- Clinical improvement in treatment with thionamides appears already by the end of the 1st week of therapy, and euthyroidism is achieved after 4-6 weeks. As a result of the well-known immunosuppressive effect of pregnancy, manifested by an increase in the number of T-suppressors and a decrease in the number of T-helpers, DTG during pregnancy tends to spontaneous remission. Knowledge of this feature of the course of thyrotoxicosis during pregnancy makes it possible to control the function of the thyroid gland of the mother with the help of relatively low initial and maintenance doses of thyreostatics. The drugs should be administered at the lowest possible initial dosage (no higher than 10-15 mg of mercazolil or 100 mg of propylthiouracil per day) with the transition to a maintenance dose (2.5 mg/day for mercazolil and 50 mg/day for propylthiouracil).
- Treatment according to the "block and replace" method with high doses of thionamides in combination with replacement therapy T 4 during pregnancy is contraindicated.
 With this regimen of therapy, T 4 maintains euthyroidism only in the mother, at the same time it can cause hypothyroidism in fetus, since high doses of thyreostatics, in contrast to T 4 , easily pass through the placenta.
With this regimen of therapy, T 4 maintains euthyroidism only in the mother, at the same time it can cause hypothyroidism in fetus, since high doses of thyreostatics, in contrast to T 4 , easily pass through the placenta. - The use of p-adrenergic antagonists during pregnancy complicated by the development of thyrotoxicosis is undesirable, since they can cause a decrease in placental mass, intrauterine growth retardation, postnatal bradycardia and hypoglycemia, and also weaken the response to hypoxia, p-blockers can only be used for a short the period for preparation for surgical treatment or with the development of a thyrotoxic crisis.
- The optimal method for monitoring the effectiveness of thyrotoxicosis treatment during pregnancy is to determine the concentration of SvTz and SvT 4 in the blood. The levels of svT 4 and svT 3 in the blood serum of the mother during treatment with thyreostatics should be maintained at the upper limit of the norm in order to avoid hypothyroidism in the fetus.
Due to the physiological changes in TSH secretion during the various phases of pregnancy, the TSH blood level is not a reliable criterion for judging the adequacy of treatment. At the same time, a very high level of TSH indicates the development of drug-induced hypothyroidism and requires immediate withdrawal or reduction of the dose of thionamides. Recommended by a number of authors, ultrasound determination of the size of the thyroid gland of the fetus and the study of the level of TSH, T 3 T 4 in the blood of the fetus, unfortunately, is available only to a small circle of highly specialized medical institutions and cannot yet be widely used.
- If there is stable compensation in the last months of pregnancy, thyreostatic drugs can be canceled. At the same time, one should be aware of the frequent recurrence of thyrotoxicosis in the postpartum period.
- During lactation, thionamides may pass into breast milk, with mercazolil to a greater extent than propylthiouracil.
 However, there is evidence that low doses of thionamides (up to 15 mg mercazolil and 150 mg propylthiouracil) taken by a woman while breastfeeding do not appear to affect the infant's thyroid function.
However, there is evidence that low doses of thionamides (up to 15 mg mercazolil and 150 mg propylthiouracil) taken by a woman while breastfeeding do not appear to affect the infant's thyroid function.
Why is it so important to treat thyrotoxicosis during pregnancy?
Pregnancy thyrotoxicosis increases the risk of stillbirth, preterm labor or preeclampsia. There is also an increase in the incidence of neonatal mortality and the likelihood of a child being born with a lack of body weight. Decompensated thyrotoxicosis can cause and aggravate cardiovascular insufficiency in the mother, as well as contribute to the development of a thyrotoxic crisis during labor pains and attempts.
It should be noted that the above complications are more often observed in the development of thyrotoxicosis during pregnancy than in the case of pregnancy in women with previously treated DTG. There is no doubt that adequate control and treatment of thyrotoxicosis in the mother are the main factor in improving the prognosis of pregnancy and childbirth.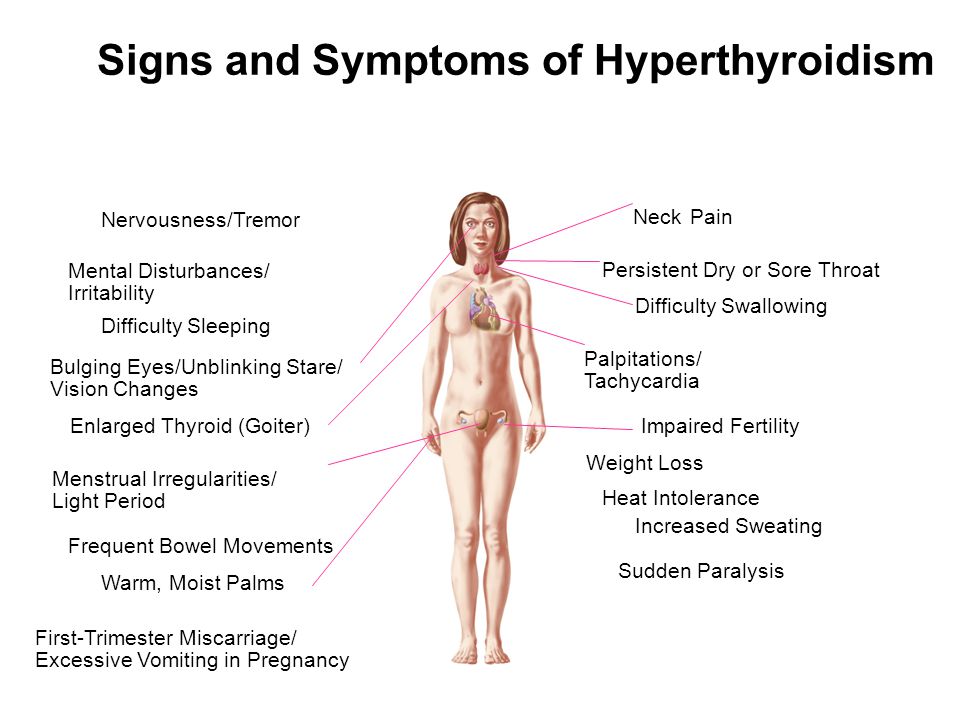
Children born to mothers with decompensated DTG have an increased risk of congenital malformations and other fetal disorders (6%). At the same time, in children whose mothers during pregnancy were in a state of drug euthyroidism during treatment with methimazole, the frequency of fetal disorders is similar to that among children of healthy euthyroid mothers (< 1%).
There is no information in the literature about the teratogenic effects of propylthiouracil, while: Methimazole is extremely rarely accompanied by a congenital malformation of the skin (aplasi cutis). Studies of the intellectual development of children exposed to thyreostatics during fetal development also did not reveal deviations from normal indicators.
All these data confirm that untreated] maternal thyrotoxicosis may cause congenital malformations of the fetus and other complications of pregnancy, and that the benefits of thyreostatic treatment outweigh any possible teratogenic effects associated with these] drugs.
Intrauterine thyrotoxicosis develops when the function of the mature thyroid gland of the fetus is stimulated by a large amount of immunoglobulins and maternal blood. This condition may develop only after about the 25th week of pregnancy. Fetal thyrotoxicosis can be established by measuring the heart rate (above 160 per minute), determining the level of TSH or integral TG level obtained by amniocentesis or cordocentesis, as well as ultrasound, which allows establish the presence of goiter in the fetus. The basis of the treatment of fetal thyrotoxicosis is its temporary administration of thyrostatic therapy to the mother, and the heart rate is reduced!!! fetus during treatment should be within 140 beats per minute. 9lasts 2-3 months, may be a placenta! passage of thyroid-stimulating immunoglobulins. The clinical symptoms of neonatal thyrotoxicosis are tachycardia, hypersensitivity, growth retardation, increased bone age, goiter (not always), premature, craniostenosis, increased mortality and morbidity.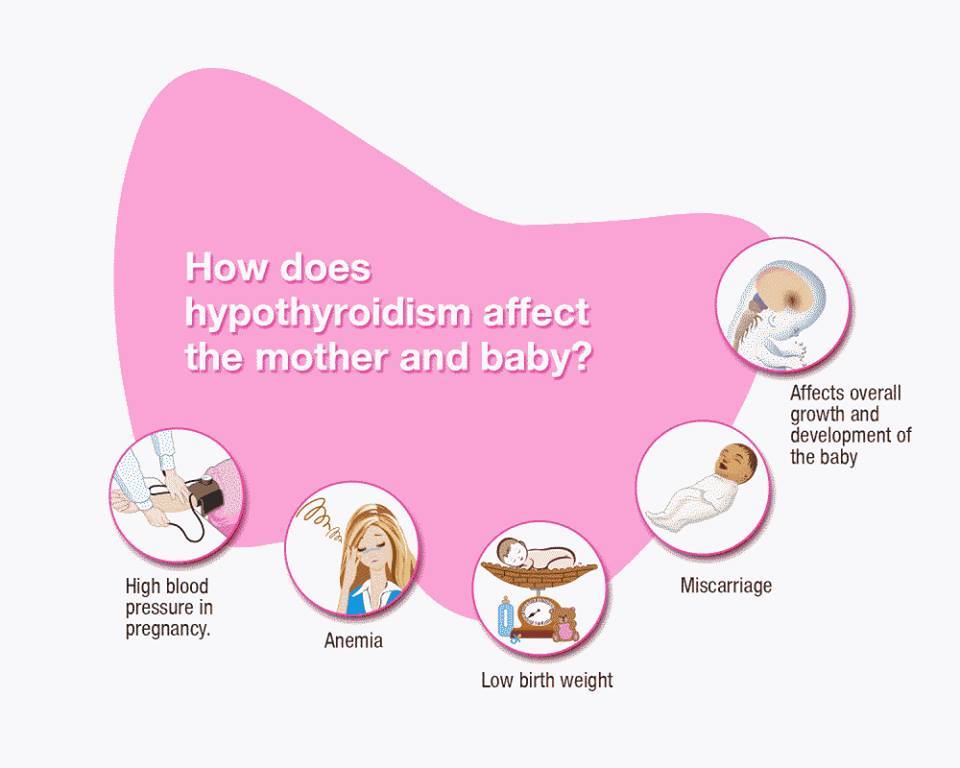
Neonatal hyperthyroidism requires the earliest possible and active treatment with thionamides. Newborns are prescribed methimazole (0.5-1 mg / kg body weight per day) or propylthiouracil (5-10 mg / kg body weight per day) in 3 divided doses. It is possible to prescribe propranolol to slow down the heart rate and reduce catecholamine activity. In severe disease, a saturated solution of iodide (1 drop of solution per day for no more than 3 weeks) can be given to inhibit the release of previously synthesized triglycerides.
In severe cases, the addition of glucocorticoids is necessary, which, in addition to the general effect, also have the ability to block the conversion of T 4 vT 3 .
The most common causes of primary hypothyroidism in pregnant women are chronic autoimmune thyroiditis (AIT) and the condition after thyroid resection for DTG and various forms of goiter. Hypothyroidism due to AIT in most cases is detected and compensated before pregnancy, but sometimes its debut coincides with pregnancy.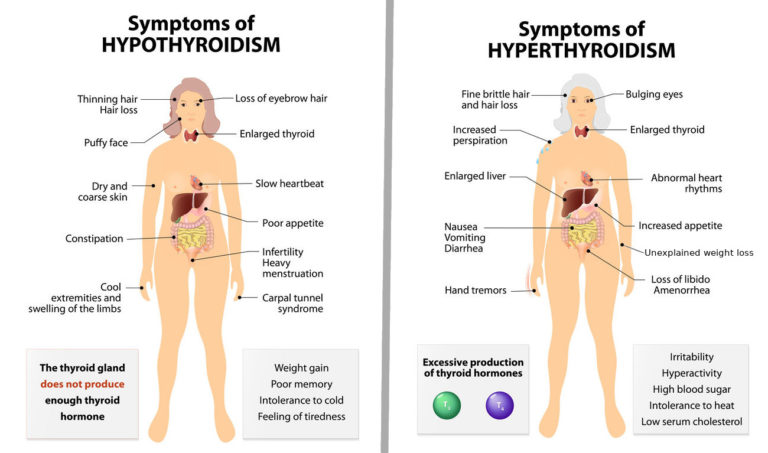
In order to detect AIT during pregnancy, it is necessary to examine pregnant women with suspected thyroid dysfunction for the presence of antibodies to thyroglobulin and thyroid peroxidase in the blood serum.
As previously described, due to the immunosuppressive effects of pregnancy, previously diagnosed AIT may tend to remit during pregnancy with relapse in the postpartum period.
The most typical symptoms of hypothyroidism during pregnancy are weakness, increased dryness of the skin, fatigue and constipation, however, it should be remembered that these symptoms can also be manifestations of pregnancy itself in the absence of a decrease in thyroid function. The diagnosis of hypothyroidism during pregnancy is made on the basis of a decrease in the level of fT 4 and increased serum TSH levels.
The selection of an adequate dose of T 4 is carried out under the control of the level of TSH and sT 4 in the blood serum (100-150 mcg of T 4 per day). Until recently, it was believed that pregnant women with previously treated hypothyroidism do not need to increase the dose of T 4 on the basis that the increased need for thyroid hormones is compensated by an increase in their concentration in blood serum and a decrease in metabolic conversion of T 4 . However, it has now become clear that women suffering from hypothyroidism and receiving T 4 replacement therapy often need to increase the dose of T 4 during pregnancy.
Until recently, it was believed that pregnant women with previously treated hypothyroidism do not need to increase the dose of T 4 on the basis that the increased need for thyroid hormones is compensated by an increase in their concentration in blood serum and a decrease in metabolic conversion of T 4 . However, it has now become clear that women suffering from hypothyroidism and receiving T 4 replacement therapy often need to increase the dose of T 4 during pregnancy.
Probable reasons for the increased need for triglycerides during pregnancy can be both an increase in body weight with increasing gestational age, and adaptive regulation of the hypothalamus-pituitary-thyroid gland, as well as possible changes in peripheral metabolism T 4 due to the presence of feto-placental complex.
Inadequate treatment of maternal hypothyroidism can lead to pregnancy complications such as anemia, preeclampsia, placental abruption, postpartum hemorrhage, and cardiovascular dysfunction. In addition, in the fetus and neonate with congenital hypothyroidism, the transplacental passage of maternal T 4 during early pregnancy may play a critical role in normal brain development.
In addition, in the fetus and neonate with congenital hypothyroidism, the transplacental passage of maternal T 4 during early pregnancy may play a critical role in normal brain development.
Blocking antibodies to TSH receptors that cross the placenta to the fetus can cause fetal and neonatal hypothyroidism (similar to fetal and neonatal hyperthyroidism). It is important to note that children of mothers suffering from hypothyroidism with the presence of antibodies that block the TSH receptor have an increased risk of developing intrauterine or postpartum hypothyroidism, even if the mother reaches the euthyroid state after T replacement therapy 4 .
Fetal hypothyroidism is accompanied by intrauterine growth retardation, bradycardia, delayed development of ossification nuclei, as well as impaired development of the fetal central nervous system.
Neonatal hypothyroidism usually lasts for 1-4 months (the half-life of maternal antibodies from the baby's blood is on average 3 weeks). The most typical signs of hypothyroidism in the early postnatal period are postterm pregnancy (gestational age > 42 weeks), high birth weight (> 4 kg), macroglossia, peripheral cyanosis and edema, difficulty breathing, low rough voice when crying and screaming. The diameter of the occipital fontanel exceeds 5 mm, the period of jaundice lengthens (> 3 days). In the future, drowsiness, decreased appetite, decreased activity, hypothermia, dryness and pallor of the skin may occur.
The most typical signs of hypothyroidism in the early postnatal period are postterm pregnancy (gestational age > 42 weeks), high birth weight (> 4 kg), macroglossia, peripheral cyanosis and edema, difficulty breathing, low rough voice when crying and screaming. The diameter of the occipital fontanel exceeds 5 mm, the period of jaundice lengthens (> 3 days). In the future, drowsiness, decreased appetite, decreased activity, hypothermia, dryness and pallor of the skin may occur.
Other causes of transient hypothyroidism may be the functional immaturity of the hypothalamic-pituitary system in premature infants, as well as severe iodine deficiency in the mother during pregnancy and the appointment of high doses of thyreostatics for DTG. The proven role of TSH in fetal development, as well as the effect of TG on growth and development in the neonatal period (especially during the 1st year of life) necessitates screening for congenital (including transient) hypothyroidism.
Transient hypothyroxinemia in most cases disappears on its own when the cause that caused it disappears. In some cases, the appointment of a newborn T 4 at a dose of 10-15 mcg / kg of body weight per day in a short course (3-4 weeks) is indicated.
In some cases, the appointment of a newborn T 4 at a dose of 10-15 mcg / kg of body weight per day in a short course (3-4 weeks) is indicated.
In the postpartum period, 4-16.7% of women with no history of thyroid disease may develop postpartum thyroiditis (PT). The etiology of this disease is still not fully understood. The revealed relationship between PT and the detection of autoantibodies to thyroid tissue (to thyroid peroxidase and microsomal antigens), the presence of certain HLA markers and lymphocytic infiltration of thyroid tissue allows us to consider PT as a type of AIT.
During the PT there is a certain phase. After an optional phase of destructive hyperthyroidism, which occurs in the form of painless asymptomatic thyroiditis (1-4th month of the postpartum period), in about 23% of cases, a phase of persistent hypothyroidism occurs (5-7th month of the postpartum period).
Clinical manifestations of hypothyroidism in PT are usually typical (weakness, dry skin, tendency to constipation, etc.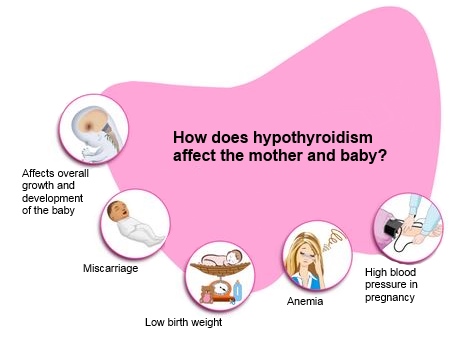 ). When scanning the thyroid gland, a reduced absorption of the radioactive isotope of iodine is noted. The ultrasound picture of PT is characterized by a diffuse or multifocal decrease in thyroid echogenicity and structural changes characteristic of lymphocytic infiltration of the thyroid gland and disorders of thyroid morphology.
). When scanning the thyroid gland, a reduced absorption of the radioactive isotope of iodine is noted. The ultrasound picture of PT is characterized by a diffuse or multifocal decrease in thyroid echogenicity and structural changes characteristic of lymphocytic infiltration of the thyroid gland and disorders of thyroid morphology.
With the development of persistent hypothyroidism, replacement therapy T 4 is prescribed according to the usual scheme.
Currently, the relationship between the presence in the postpartum period of an increased titer of antibodies to thyroid tissue and postpartum depression is being investigated. It is assumed that these antibodies can modulate the function of neurotransmitters, as well as affect cytokine receptors in the brain.
In conclusion, I would like to note that timely and adequate treatment of thyroid diseases in pregnant women contributes both to the normal course of pregnancy and to the correct physical and intellectual development of the child.
:strip_icc():format(jpeg)/kly-media-production/medias/2785562/original/028627600_1556001360-shutterstock_1019963743.jpg)