What causes babies to be born blind
Severe Visual Impairment and Blindness in Infants: Causes and Opportunities for Control
Middle East Afr J Ophthalmol. 2011 Apr-Jun; 18(2): 109–114.
doi: 10.4103/0974-9233.80698
,1,2,3 and 4
Author information Copyright and License information Disclaimer
Childhood blindness has an adverse effect on growth, development, social, and economic opportunities. Severe visual impairment (SVI) and blindness in infants must be detected as early as possible to initiate immediate treatment to prevent deep amblyopia. Although difficult, measurement of visual acuity of an infant is possible. The causes of SVI and blindness may be prenatal, perinatal, and postnatal. Congenital anomalies such as anophthalmos, microphthalmos, coloboma, congenital cataract, infantile glaucoma, and neuro-ophthalmic lesions are causes of impairment present at birth. Ophthalmia neonatorum, retinopathy of prematurity, and cortical visual impairment are acquired during the perinatal period. Leukocoria or white pupillary reflex can be cause by congenital cataract, persistent hyperplastic primary vitreous, or retinoblastoma. While few medical or surgical options are available for congenital anomalies or neuro-ophthalmic disorders, many affected infants can still benefit from low vision aids and rehabilitation. Ideally, surgery for congenital cataracts should occur within the first 4 months of life. Anterior vitrectomy and primary posterior capsulotomy are required, followed by aphakic glasses with secondary intraocular lens implantation at a later date. The treatment of infantile glaucoma is surgery followed by anti-glaucoma medication. Retinopathy of prematurity is a proliferation of the retinal vasculature in response to relative hypoxia in a premature infant. Screening in the first few weeks of life can prevent blindness. Retinoblastoma can be debulked with chemotherapy; however, enucleation may still be required. Neonatologists, pediatricians, traditional birth attendants, nurses, and ophthalmologists should be sensitive to a parent’s complaints of poor vision in an infant and ensure adequate follow-up to determine the cause.
 If required, evaluation under anesthesia should be performed, which includes funduscopy, refraction, corneal diameter measurement, and measurement of intraocular pressure.
If required, evaluation under anesthesia should be performed, which includes funduscopy, refraction, corneal diameter measurement, and measurement of intraocular pressure.
Keywords: Childhood Blindness, Congenital Anomalies, Congenital Cataract, Retinopathy of Prematurity
Visual impairment and blindness in children pose a special problem for ophthalmologists, as many eye care practitioners are not familiar with performing pediatric eye examinations and measuring visual acuity in infants. Infants are unable to verbalize their complaints, and history from parents and care takers may lack important details. The first year of life is also the time when the visual system develops and binocular vision is formed.1 If a visual deficit at this age is not treated in a timely manner, amblyopia and permanent visual deficit can occur. Hence, early diagnosis and prompt treatment is essential. The burden of blindness measured in blind-person years due to childhood blindness is second only to cataract – the most common cause of avoidable blindness in childhood. 2 Studies worldwide show that many of the causes of blindness in children are either preventable or treatable (ie, avoidable).3 Even children who have visual loss that cannot be clinically treated, can be helped with low vision devices and rehabilitation. Childhood blindness affects the individual, their family, and the community. Blindness also has implications for infants’ development, education, and future social, marital, and economic prospects. Nearly 75% of early learning comes from vision. Early onset visual loss can have profound consequences on a child’s motor, social, emotional, and psychological development.4
2 Studies worldwide show that many of the causes of blindness in children are either preventable or treatable (ie, avoidable).3 Even children who have visual loss that cannot be clinically treated, can be helped with low vision devices and rehabilitation. Childhood blindness affects the individual, their family, and the community. Blindness also has implications for infants’ development, education, and future social, marital, and economic prospects. Nearly 75% of early learning comes from vision. Early onset visual loss can have profound consequences on a child’s motor, social, emotional, and psychological development.4
The World Health Organization (WHO) classifies the causes of childhood blindness according to the anatomical site most affected and the underlying etiology.5 Visual loss in infants can be either prenatal (ie, occurring at the time of conception or during the intrauterine period) or postnatal (during or after birth). Prenatal causes are congenital anomalies – anophthalmos, microphthalmos, and coloboma; congenital cataract, retinal dystrophies such as Leber’s congenital amaurosis, infantile glaucoma, and congenital cloudy cornea. In the perinatal period (ie, from the 28th week of gestation through to 1-4 weeks after birth), the following conditions can occur: cortical impairment from birth asphyxia, ophthalmia neonatorum, and retinopathy of prematurity. Postnatal conditions (ie, those acquired after birth) are unusual during infancy. As retinoblastoma can be inherited as an autosomal dominant condition, it is considered a prenatal condition for the purposes of this paper.
In the perinatal period (ie, from the 28th week of gestation through to 1-4 weeks after birth), the following conditions can occur: cortical impairment from birth asphyxia, ophthalmia neonatorum, and retinopathy of prematurity. Postnatal conditions (ie, those acquired after birth) are unusual during infancy. As retinoblastoma can be inherited as an autosomal dominant condition, it is considered a prenatal condition for the purposes of this paper.
Leukocoria may be seen in early infancy can be due to congenital cataract, retinopathy of prematurity (ROP), persistent hyperplastic primary vitreous (PHPV) – anterior or posterior – retinoblastoma or rare genetic disorders such as Norrie’s disease. A leukocoria detected by parents in later infancy may be due to developmental cataract or retinoblastoma. Retinoblastoma is the most common childhood tumor presenting at the age of 12-15 months in bilateral cases and 18-24 months in unilateral cases; it can also develop much later in childhood.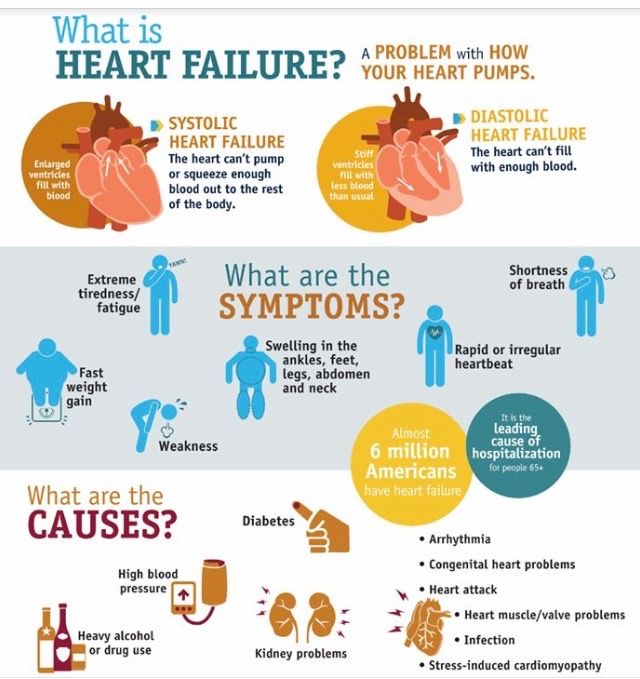 It is essential to examine children presenting with leukocoria under anesthesia due to the potentially serious underlying causes.
It is essential to examine children presenting with leukocoria under anesthesia due to the potentially serious underlying causes.
Congenital anomalies
Anophthalmos and microphthalmos are congenital anomalies in which the entire eyeball is entirely absent or smaller than normal from birth. Typical complete uveal coloboma is the most common congenital anomaly in which the embryonic fissure does not close completely by the sixth week of intrauterine life. Coloboma can cause severe visual loss if the defect involves the macula and/or optic nerve. Surveys from schools for the blind in many parts of Asia show that these congenital eye anomalies are a leading cause of blindness and severe visual impairment in children.6–9 These are difficult to research, even in countries with the best healthcare systems and expertise. In the majority of cases, even with in-depth molecular genetic testing and detailed investigation, no cause can be identified.10 This is because these anomalies are likely to be due to genes controlling eye development (which are largely unknown) and possibly, gene-environment interactions, reflecting similar processes elucidated for folic acid and spina bifida. 11 Even if an environmental agent (eg, folate) could be identified, the intervention would have to been extremely broad to have a measurable effect, be inexpensive and safe (as it would need to be given to large numbers of people) and be nonteratogenic – a difficult proposition in developing countries.
11 Even if an environmental agent (eg, folate) could be identified, the intervention would have to been extremely broad to have a measurable effect, be inexpensive and safe (as it would need to be given to large numbers of people) and be nonteratogenic – a difficult proposition in developing countries.
Medical therapy or surgery are rarely effective in congenital cases; however, refraction and low vision aids can help many of these cases.12 Many children with congenital defects can be taught to read print with low vision aids instead of Braille.
PHPV is a congenital anomaly in which the primary vitreous, which extends from the optic disc to the vitreous base at the posterior pole of the lens, persists into childhood.13 This anomaly can cause severe visual loss, and may be associated with microphthalmos. PHPV is usually unilateral, although bilateral cases can occur. Early vitrectomy can be attempted in eyes where the surrounding vitreous is clear and the retina fairly developed; however, the visual outcomes are often suboptimal. 14
14
Infantile glaucoma
Infantile glaucoma can result in blindness without early intervention.15 Surgery followed by medication and optical aids is the treatment of choice. Numerous genes have been implicated and genetic counselling is essential if more than one sibling is affected.
Retinal dystrophies
Retinal dystrophies have variable genetic inheritance and are common in communities with high rates of consanguinity. Although the dystrophy may only give visual symptoms later in life, this is a common cause of blindness in children worldwide, particularly in Middle Eastern countries.16,17 Low vision aids are effective for navigation and reading for individuals affected with retinal dystrophies. Genetic counselling is required if more than one sibling is involved.
Leber’s congenital amaurosis is an autosomal recessive condition that manifests in infancy. The fundus may be normal or show peripheral chorioretinal atrophy and granularity with nystagmus. Oculodigital syndrome, in which the infant constantly rubs or presses on the eyes, which leads to enophthalmos, is common. Other common features include learning disability, deafness, and epilepsy. In Knobloch syndrome nystagmus, high myopia and late retinal detachment are seen. Oculocutaneous albinism, which may be X-linked or autosomal recessive, is detectable at birth. Ocular albinism affects only male infants due to the X-linked inheritance and is usually diagnosed at infancy. Achromatopsy is a rare autosomal recessive heterogeneous group of stationary retinal dystrophies characterized by reduced central vision, poor color vision, and normal fundus. The usual presentation is reduced vision, marked photophobia, and nystagmus in infancy. A macular cherry red spot could be due to Tay-Sachs disease, which is fatal by the second year of life or due to Niemann-Pick disease or Sandhoff disease.
Oculodigital syndrome, in which the infant constantly rubs or presses on the eyes, which leads to enophthalmos, is common. Other common features include learning disability, deafness, and epilepsy. In Knobloch syndrome nystagmus, high myopia and late retinal detachment are seen. Oculocutaneous albinism, which may be X-linked or autosomal recessive, is detectable at birth. Ocular albinism affects only male infants due to the X-linked inheritance and is usually diagnosed at infancy. Achromatopsy is a rare autosomal recessive heterogeneous group of stationary retinal dystrophies characterized by reduced central vision, poor color vision, and normal fundus. The usual presentation is reduced vision, marked photophobia, and nystagmus in infancy. A macular cherry red spot could be due to Tay-Sachs disease, which is fatal by the second year of life or due to Niemann-Pick disease or Sandhoff disease.
Congenital cataract
The birth prevalence of cataract is 3-5/1,000 live births.18 While monogenetic abnormalities, syndromes, metabolic disorders, genetic disorders, and congenital rubella syndrome are recognized causes of this condition, in the majority, the cause is unknown. 19 The importance of congenital cataract is increasing as a cause of blindness among children in developing countries, with other causes declining. There can be 2 years delay from cataract diagnosis to presentation for surgery.20 Early recognition and referral is essential to prevent development of dense amblyopia. However, the timing of intraocular lens (IOL) implantation remains controversial.21 Many experienced surgeons are implanting IOLs in very young children, ie, 12 months or older. While children with unilateral cataract have undergone IOL implantation at as early as 6 months of age, it is better to wait until 2 years of age for implanting IOLs in children with bilateral cataracts.21,22 In \ children where the risk of general anesthesia very high, bilateral surgery at the same visit is advisable; however, this should be an exception rather than the rule. There is almost a hundred percent posterior capsular opacification in pediatric eyes.
19 The importance of congenital cataract is increasing as a cause of blindness among children in developing countries, with other causes declining. There can be 2 years delay from cataract diagnosis to presentation for surgery.20 Early recognition and referral is essential to prevent development of dense amblyopia. However, the timing of intraocular lens (IOL) implantation remains controversial.21 Many experienced surgeons are implanting IOLs in very young children, ie, 12 months or older. While children with unilateral cataract have undergone IOL implantation at as early as 6 months of age, it is better to wait until 2 years of age for implanting IOLs in children with bilateral cataracts.21,22 In \ children where the risk of general anesthesia very high, bilateral surgery at the same visit is advisable; however, this should be an exception rather than the rule. There is almost a hundred percent posterior capsular opacification in pediatric eyes. Experts recommend primary posterior capsulotomy and anterior vitrectomy in children up to the age of 6-7 years. At 6 years or older, the child can sit on a slit lamp for Nd:YAG laser capsulotomy after surgery, if required.21 The anterior vitrectomy removes the almost solid vitreous base, which can act as a scaffold for migrating lenticular cells. The pars plana approach has the advantage of keeping the primary incision and the anterior chamber free of vitreous, but this is technically demanding for anterior segment surgeons. Moreover, there is an exaggerated fear of suprachoroidal hemorrhage.23 Vitrectomy through limbal side ports is easier but the surgeon has to be meticulous in removing vitreous from the anterior chamber. The primary posterior capsulotomy can be performed before or after inserting the intraocular lens. There is a risk of the capsulotomy extending during the implantation of a foldable lens. An MVR blade may be used to make a ’nick’ below the implanted lens to later enlarge the opening with a vitrectors.
Experts recommend primary posterior capsulotomy and anterior vitrectomy in children up to the age of 6-7 years. At 6 years or older, the child can sit on a slit lamp for Nd:YAG laser capsulotomy after surgery, if required.21 The anterior vitrectomy removes the almost solid vitreous base, which can act as a scaffold for migrating lenticular cells. The pars plana approach has the advantage of keeping the primary incision and the anterior chamber free of vitreous, but this is technically demanding for anterior segment surgeons. Moreover, there is an exaggerated fear of suprachoroidal hemorrhage.23 Vitrectomy through limbal side ports is easier but the surgeon has to be meticulous in removing vitreous from the anterior chamber. The primary posterior capsulotomy can be performed before or after inserting the intraocular lens. There is a risk of the capsulotomy extending during the implantation of a foldable lens. An MVR blade may be used to make a ’nick’ below the implanted lens to later enlarge the opening with a vitrectors. 23 A-scan biometry and keratometry need to be performed under anesthesia. Hydrophobic acrylic or poly-methyl-methacrylate IOLs are best for infants. Generally, the target postoperative refraction is emmetropia by the age of 5-6 years. Aphakic glasses have to be corrected for near vision.
23 A-scan biometry and keratometry need to be performed under anesthesia. Hydrophobic acrylic or poly-methyl-methacrylate IOLs are best for infants. Generally, the target postoperative refraction is emmetropia by the age of 5-6 years. Aphakic glasses have to be corrected for near vision.
Pediatric cataract surgery is one step in a series of interventions required to rehabilitate vision.24 Postoperative management includes frequent steroid eye drops with cycloplegia, followed by early, accurate, and repeated refractions and treatment of amblyopia by patching the better seeing eye. Late complications of pediatric IOL implantation include opacification of the visual axis and glaucoma, the latter being particularly difficult to treat. Parents must be counselled about the need for regular ophthalmic examination, refraction, and intraocular pressure measurement. It is important to remember that aphakic/pseudophakic children need correction for near as well as distance, and many are able to tolerate executive bifocals very well. 21,24
21,24
Retinoblastoma
Retinoblastoma is the most common intraocular malignancy in early childhood. It usually presents after infancy as leukocoria, esotropia, or masquerades as uveitis. Lesions detected early can be treated with chemotherapy with preservation of the globe; however, larger lesions may need enucleation.25,26 A prosthesis needs to be implanted in the child’s eye to prevent the contracture of the socket. A mule’s implant wrapped in a sclera shell or dermal fat grafts may be used for this purpose. Management with specialist tertiary oncology services is also advisable.
Ophthalmia neonatorum
This is an eminently preventable condition in which the eyes of the infant become infected in the birth passage during delivery. Credé’s prophylaxis and antenatal testing or treatment of sexually transmitted diseases during pregnancy can prevent the condition. Credé’s prophylaxis entails cleaning the infants’ eyes immediately after birth with instillation of a topical antibiotic (eg, tetracycline eye ointment) or antiseptic (eg, 2. 5% povidone iodine). Traditionally, a 2% solution of silver nitrate was used for Credé’s prophylaxis. The agent used depends on the local epidemiology of sexually transmitted diseases and the organism’s sensitivity to antibiotics. Treatment of ophthalmia neonatorum must entail systemic as well as topical treatment, to ensure treatment of extraocular sites of infection (eg, pneumonia).
5% povidone iodine). Traditionally, a 2% solution of silver nitrate was used for Credé’s prophylaxis. The agent used depends on the local epidemiology of sexually transmitted diseases and the organism’s sensitivity to antibiotics. Treatment of ophthalmia neonatorum must entail systemic as well as topical treatment, to ensure treatment of extraocular sites of infection (eg, pneumonia).
Retinopathy of prematurity
Retinopathy of prematurity (ROP) is responsible for up to 15% of all causes of blindness in developed countries and up to 60% in middle income countries.27 A number of risk factors are implicated in the development of ROP. However, the most commonly identified risk factors are the degree of immaturity measured by birth weight (BW), gestational age (GA), and prolonged exposure to supplementary oxygen.28 Paradoxically, the hyperbaric oxygen that saves an infant’s life, if not properly controlled and tapered, can cause blindness. There are several trials seeking to define safe upper and lower limits of arterial oxygen saturation. 29 The improvement in neonatal care can lead to an increase in survival rates of premature infants in middle-income countries and major metropolises of even poorer countries. As a consequence, ROP has become a very important cause of childhood in these developing economies, where quality of neonatal care still needs to improve. The disease is classified by severity (stages I-V), by site (zones 1-3), and by extent (clock hours 1-12). While most developed countries screen infants by BW < 1,500 g,30 middle income and developing countries need larger BW criteria as infants weighing up to 2,500 g are developing severe ROP.31,32 Appropriate screening protocols ensure early detection in the first weeks of life so that treatment (peripheral ablation of avascular retina) with laser or cryotherapy can be provided. So far, there is no evidence provided by clinical trials that surgery is beneficial for cases in stages IV and V. Anti-vascular endothelial growth factors (VEGFs) are promising drugs for treatment of retinal proliferative diseases in adults.
29 The improvement in neonatal care can lead to an increase in survival rates of premature infants in middle-income countries and major metropolises of even poorer countries. As a consequence, ROP has become a very important cause of childhood in these developing economies, where quality of neonatal care still needs to improve. The disease is classified by severity (stages I-V), by site (zones 1-3), and by extent (clock hours 1-12). While most developed countries screen infants by BW < 1,500 g,30 middle income and developing countries need larger BW criteria as infants weighing up to 2,500 g are developing severe ROP.31,32 Appropriate screening protocols ensure early detection in the first weeks of life so that treatment (peripheral ablation of avascular retina) with laser or cryotherapy can be provided. So far, there is no evidence provided by clinical trials that surgery is beneficial for cases in stages IV and V. Anti-vascular endothelial growth factors (VEGFs) are promising drugs for treatment of retinal proliferative diseases in adults. However, extreme caution is warranted in infants, as these drugs interfere with the normal development of vasculature elsewhere in the body and may impede development of the lungs and alveoli of the lungs, which are VEGF-sensitive and develop at the same time as does ROP.33 Premature infants are at increased risk of developing refractive errors, strabismus, amblyopia, and low vision; hence, long-term follow-up is recommended.
However, extreme caution is warranted in infants, as these drugs interfere with the normal development of vasculature elsewhere in the body and may impede development of the lungs and alveoli of the lungs, which are VEGF-sensitive and develop at the same time as does ROP.33 Premature infants are at increased risk of developing refractive errors, strabismus, amblyopia, and low vision; hence, long-term follow-up is recommended.
Optic nerve lesions and cerebral visual impairment
Optic nerve lesions and cerebral visual impairment are the most common causes of visual impairment in many developed countries34 where these are often a consequence of preterm birth.35 Birth asphyxia, which causes cerebral palsy, may affect the optic nerve and cause cortical visual impairment. Little can be done by way of medical treatment. Low vision aids and rehabilitation are often the only recourse. Often, such children are severely handicapped, which makes assessment and management even more challenging.
Keratomalacia cause by acute deficiency of vitamin A is very unusual in the first year of life, and most infants before the age of 9 months (when they are immunized) will have protective measles antibodies received from their mother. The common causes of blindness in childhood such as vitamin A deficiency, measles, trauma, and trachoma are uncommon in infancy. Unless the refractive error is very large [>3-4 diopter (D)], or is causing strabismus, spectacles are not required in infants. However, if the infant has low vision, even small refractive errors may need correction.
While evaluation of a visually impaired infant may seem challenging for the general ophthalmologist, the task is not that difficult. Infants are best examined in the position they are most comfortable – on their parents’ shoulders, in their laps, or cribs. We recommend not making the infant and parents wait in a busy waiting area. A separate area for breastfeeding is welcome, as this puts the mother and child at ease. The infant can be examined for fixating and following of the flashlight. The ophthalmologist should also have a quick look at the cornea, anterior chamber, lens, and pupillary reflex. A handheld slit lamp is the best instrument for this component of the evaluation. The red reflex should be examined to rule out congenital cataract, advanced ROP, and other causes of leukocoria. If the child is uncooperative or distressed, the examination can be repeated after a few hours or next day after instilling atropine eye ointment to dilate the pupil. However, the drug may affect the pupil for a week. Cyclopentolate 0.1%, tropicamide 0.1%, and phenylephrine 2.5% eye drops may also be instilled, though there is a risk of toxicity due to systemic absorption. Cyclopentolate and tropicamide are parasympatholytic agents acting on sphincter pupillae, whereas phenylephrine is sympathomimetic acting on the dilator papillae muscle. The child can also be given 2.5 ml of phenergan syrup (properazine – a mild sedative and antihistamine combination) and then breastfed to lull it into sleep.
The infant can be examined for fixating and following of the flashlight. The ophthalmologist should also have a quick look at the cornea, anterior chamber, lens, and pupillary reflex. A handheld slit lamp is the best instrument for this component of the evaluation. The red reflex should be examined to rule out congenital cataract, advanced ROP, and other causes of leukocoria. If the child is uncooperative or distressed, the examination can be repeated after a few hours or next day after instilling atropine eye ointment to dilate the pupil. However, the drug may affect the pupil for a week. Cyclopentolate 0.1%, tropicamide 0.1%, and phenylephrine 2.5% eye drops may also be instilled, though there is a risk of toxicity due to systemic absorption. Cyclopentolate and tropicamide are parasympatholytic agents acting on sphincter pupillae, whereas phenylephrine is sympathomimetic acting on the dilator papillae muscle. The child can also be given 2.5 ml of phenergan syrup (properazine – a mild sedative and antihistamine combination) and then breastfed to lull it into sleep. This allows more detailed fundus and external ocular evaluation, and measurement of intraocular pressure by non-contact or Perkins (more accurate) tonometer. If a serious lesion such as retinoblastoma is suspected, an evaluation under anesthesia is essential for detailed examination of the fundus. The opportunity must be utilized not just to examine the fundus but to also perform tonometry, refraction, measurement of corneal diameter, and axial length.
This allows more detailed fundus and external ocular evaluation, and measurement of intraocular pressure by non-contact or Perkins (more accurate) tonometer. If a serious lesion such as retinoblastoma is suspected, an evaluation under anesthesia is essential for detailed examination of the fundus. The opportunity must be utilized not just to examine the fundus but to also perform tonometry, refraction, measurement of corneal diameter, and axial length.
Orthoptic evaluation of an infant requires patience. Jampolsky’s dictum of “one toy-one look” should be used to examine the infant’s ocular motility in the nine cardinal directions of gaze. An assortment of soft toys, brightly colored objects, and even mobile phones can be used to arouse the child’s interest. The objects should not make noise as the child will be attracted through the auditory and not the visual signals, which would defeat the purpose of the examination. Visual acuity can be formally assessed using forced preferential looking tests (eg, Cardiff cards), with the child sitting comfortably on a parent’s lap. The test is based on the psychological percept that humans are attracted to novel stimuli. If the child is shown a line drawing at one end of the card sheet and the other end is kept blank, the child will divert its eyes to the drawing rather than the blank area. The line drawings are made progressively finer to estimate higher orders of form vision. Worth’s ivory fall test and small sweets commonly used to decorate cakes, “hundreds and thousands”, can also be used. The child picks up the small sweets if it is able to see them. At first, bilateral vision is tested followed by monocular testing. The mother should be asked to occlude the infant’s eyes one at a time, and report on the infant’s response. If the infant objects to occlusion of one eye, this may indicate the visual acuity of the other eye is poor.
The test is based on the psychological percept that humans are attracted to novel stimuli. If the child is shown a line drawing at one end of the card sheet and the other end is kept blank, the child will divert its eyes to the drawing rather than the blank area. The line drawings are made progressively finer to estimate higher orders of form vision. Worth’s ivory fall test and small sweets commonly used to decorate cakes, “hundreds and thousands”, can also be used. The child picks up the small sweets if it is able to see them. At first, bilateral vision is tested followed by monocular testing. The mother should be asked to occlude the infant’s eyes one at a time, and report on the infant’s response. If the infant objects to occlusion of one eye, this may indicate the visual acuity of the other eye is poor.
There is a narrow window of opportunity in treating a visually impaired infant. Binocular single vision develops by 6 months of life and a visual deficit, if not detected and treated in time, may leave the child bereft of stereopsis. The amblyopia that develops from visual deprivation of early onset, irrespective of the cause, can be dense and difficult to treat.
The amblyopia that develops from visual deprivation of early onset, irrespective of the cause, can be dense and difficult to treat.
Pediatricians, general practitioners, and midwives should be educated and encouraged to perform the red reflex test. Using the direct ophthalmoscope, they should be taught to detect any opacity seen in the infant’s red reflex. All healthcare personnel working for the care of the infant should be sensitized to the eye conditions in infancy and on the causes of childhood blindness and visual impairment. Their training curricula should emphasize on the importance of early detection and treatment of such children. Training of midwives, traditional birth attendants, healthcare workers working for child health and immunization would be of immense help in early detection of such children.
The major barrier to accessing eye care services for infants is the absence of trained personnel who can diagnose a problem early. Developed countries have established referral systems between family practitioners, health visitors, neonatal units, and pediatric ophthalmology and retina specialist. However, pediatric eye care centers are rare in developing countries. There are financial and geographic barriers for many parents from poorer communities. Diagnostic, curative, and rehabilitative services may not be available in the region. Pediatric eye care interventions are also more expensive than adult treatment. For example, general anesthesia is often required for examination and treatment. This entails liaison with other medical practitioners such as pediatricians, anesthetists, and neonatologists. Another significant barrier is the lack of knowledge on the part of healthcare providers. Many healthcare workers, including physicians ask parents to wait until the child is older before any treatment can be given. Some physicians are unsure about what needs to be done. Others believe that the problem may resolve, as the child grows older or that a unilateral condition does not matter as long as the other eye is healthy. Due to these misconceptions, appropriate referral may too late if dense amblyopia has occurred.
However, pediatric eye care centers are rare in developing countries. There are financial and geographic barriers for many parents from poorer communities. Diagnostic, curative, and rehabilitative services may not be available in the region. Pediatric eye care interventions are also more expensive than adult treatment. For example, general anesthesia is often required for examination and treatment. This entails liaison with other medical practitioners such as pediatricians, anesthetists, and neonatologists. Another significant barrier is the lack of knowledge on the part of healthcare providers. Many healthcare workers, including physicians ask parents to wait until the child is older before any treatment can be given. Some physicians are unsure about what needs to be done. Others believe that the problem may resolve, as the child grows older or that a unilateral condition does not matter as long as the other eye is healthy. Due to these misconceptions, appropriate referral may too late if dense amblyopia has occurred.
Even when the infant has been referred for treatment appropriately, many parents believe that their infant is too small to undergo surgery or wear spectacles and, in some communities, visual loss in infants is not considered a priority for the family, especially for females.
However, a child who cannot be helped by medicine or surgery may still benefit from use of spectacles and/or low vision aids. Completely and irreversibly blind children can benefit by rehabilitation and special school education. This should be emphasized during parent counselling. Ophthalmologists should be nonjudgmental and empathetic while breaking the news about a potentially blinding condition. Ocular conditions, which have a genetic or familial basis, may be blamed on one of the parents. The ophthalmologist should spend some time with the parents to reassure, counsel, and suggest various alternatives that are available for the infant. It may take the parents some time to come to terms with their child’s visual loss. Frequent ophthalmic visits to confirm the level of vision and make a definitive diagnosis can be difficult for parents. A social worker or counsellor can establish a rapport with the parents or caretakers to ensure that they comply with the treatment as much as possible.
Frequent ophthalmic visits to confirm the level of vision and make a definitive diagnosis can be difficult for parents. A social worker or counsellor can establish a rapport with the parents or caretakers to ensure that they comply with the treatment as much as possible.
Blindness and severe visual impairment in infants is not that difficult to detect and diagnose. With proper care, most of these infants can be helped and formation of dense amblyopia prevented. Even if the ophthalmologist may not be able to help medically or surgically, optical aids and rehabilitation can help children reach their full capacity.
Source of Support: Nil
Conflict of Interest: None declared.
1. Day S. Normal and abnormal visual development. In: David T, editor. Pediatric ophthalmology. Section 1. Chapter 2. Oxford UK: Blackwell Science; 1997. pp. 13–28. [Google Scholar]
2. Rahi JS, Gilbert CE, Foster A, Minassian D. Measuring the burden of childhood blindness. Br J Ophthalmol. 1999;83:387–8. [PMC free article] [PubMed] [Google Scholar]
Br J Ophthalmol. 1999;83:387–8. [PMC free article] [PubMed] [Google Scholar]
3. Gilbert C, Rahi JS, Quinn GE. Visual impairment and blindness in children. In: Jhonson GJ, Minassian D, Weale RA, West SK, editors. Epidemiology of Eye Disease. 2nd ed. London: Arnold publishers; 2003. pp. 260–86. [Google Scholar]
4. Dale N, Salt A. Early support developmental journal for children with visual impairment: The case for a new developmental framework for early intervention. Child Care Health Dev. 2007;33:684–90. [PubMed] [Google Scholar]
5. Gilbert C, Foster A, Negrel AD, Thylefors B. Childhood blindness: A new form for recording causes of visual loss in children. Bull World Health Organ. 1993;71:485–9. [PMC free article] [PubMed] [Google Scholar]
6. Gogate P, Deshpande M, Sudrik S, Kishore H, Taras S, Deshpande M. Changing pattern of childhood blindness in Maharashtra, India. Br J Ophthalmol. 2007;91:8–12. [PMC free article] [PubMed] [Google Scholar]
7. Njuguna M, Msukwa G, Shilio B, Tumwesigye C, Courtright P, Lewallen S. Causes of severe visual impairment and blindness in children in schools for the blind in eastern Africa: Changes in the last 14 years. Ophthalmic Epidemiol. 2009;16:151–5. [PubMed] [Google Scholar]
Causes of severe visual impairment and blindness in children in schools for the blind in eastern Africa: Changes in the last 14 years. Ophthalmic Epidemiol. 2009;16:151–5. [PubMed] [Google Scholar]
8. Gogate P, Kishore H, Dole K, Shetty J, Gilbert C, Ranade S, et al. The pattern of childhood blindness in Karnataka, South India. Ophthalmic Epidemiol. 2009;16:212–7. [PubMed] [Google Scholar]
9. Sitorus RS, Abidin MS, Prihartono J. Causes and temporal trends of childhood blindness in Indonesia: Study at schools for the blind in Java. Br J Ophthalmol. 2007;91:1109–13. [PMC free article] [PubMed] [Google Scholar]
10. Shah SP, Taylor AE, Sowden JC, Ragge NK, Russell-Eggitt I, Rahi JS, et al. Anophthalmos, micophthalmos and typical coloboma in the United Kingdom: A prospective study of incidence and risk. Invest Ophthalmol Vis Sci. 2011;52:558–64. [PubMed] [Google Scholar]
11. Hornby SJ, Ward SJ, Gilbert CE. Hypothesis: Eye birth defects in humans may be caused by a recessively-inherited genetic predisposition to the effects of maternal vitamin A deficiency during pregnancy. Med Sci Monit. 2003;9:HY23–6. [PubMed] [Google Scholar]
Med Sci Monit. 2003;9:HY23–6. [PubMed] [Google Scholar]
12. Hornby SJ, Adolph S, Gothwal VK, Gilbert CE, Dandona L, Foster A. Requirements for optical services in children with microphthalmos, coloboma and microcornea in southern India. Eye. 2000;14:219–24. [PubMed] [Google Scholar]
13. Shastry BS. Persistent hyperplastic primary vitreous: Congenital malformation of the eye. Clin Exp Ophthalmol. 2009;37:884–90. [PubMed] [Google Scholar]
14. Walsh MK, Drenser KA, Capone A, Jr, Trese MT. Early vitrectomy effective for bilateral combined anterior and posterior persistent fetal vasculature syndrome. Retina. 2010;30:S2–8. [PubMed] [Google Scholar]
15. Mandal AK, Bhatia PG, Bhaskar A, Nutheti R. Long-term surgical and visual outcomes in Indian children with developmental glaucoma operated on within 6 months of birth. Ophthalmology. 2004;111:283–90. [PubMed] [Google Scholar]
16. Kotb AA, Hammouda EF, Tabbara KF. Childhood blindness at a school for the blind in Riyadh, Saudi Arabia. Ophthalmic Epidemiol. 2006;13:1–5. [PubMed] [Google Scholar]
Ophthalmic Epidemiol. 2006;13:1–5. [PubMed] [Google Scholar]
17. Cetin E, Yaman A, Berk AT. Etiology of childhood blindness in Izmir, Turkey. Eur J Ophthalmol. 2004;14:531–7. [PubMed] [Google Scholar]
18. Rahi JS, Dezateux C. British Congenital Cataract Interest Group. Measuring and interpreting the incidence of congenital ocular anomalies: Lessons from a national study of congenital cataract in the UK. Invest Ophthalmol Vis Sci. 2001;42:1444–8. [PubMed] [Google Scholar]
19. Rahi JS, Dezateux C. Congenital and infantile cataract in the United Kingdom: Underlying or associated factors. British Congenital Cataract Interest Group. Invest Ophthalmol Vis Sci. 2000;41:2108–14. [PubMed] [Google Scholar]
20. Mwende J, Bronsard A, Mosha M, Bowman R, Geneau R, Courtright P. Delay in presentation to hospital for surgery for congenital and developmental cataract in Tanzania. Br J Ophthalmol. 2005;89:1478–82. [PMC free article] [PubMed] [Google Scholar]
21. Wilson ME, Pandey SK, Thakur J. Paediatric cataract blindness in the developing world: Surgical techniques and intraocular lenses in the new millennium. Br J Ophthalmol. 2003;87:14–9. [PMC free article] [PubMed] [Google Scholar]
Paediatric cataract blindness in the developing world: Surgical techniques and intraocular lenses in the new millennium. Br J Ophthalmol. 2003;87:14–9. [PMC free article] [PubMed] [Google Scholar]
22. Ram J, Brar GS, Kaushik S, Sukhija J, Bandyopadhyay S, Gupta A. Primary intraocular lens implantation in the first two years of life: safety profile and visual results. Indian J Ophthalmol. 2007;55:185–9. [PubMed] [Google Scholar]
23. Gogate P. Complications of pediatric cataract surgery. Highlights Ophthalmol. 2009;37:12–5. [Google Scholar]
24. Gogate P, Khandekar R, Srisimal M, Dole K, Taras S, Kulkarni S, et al. Cataracts with delayed presentation. Are they worth operating upon? Ophthalmic Epidemiol. 2010;17:25–33. [PubMed] [Google Scholar]
25. Shields CL, Honavar SG, Meadows AT, Shields JA, Demirci H, Naduvilath TJ. Chemoreduction for unilateral retinoblastoma. Arch Ophthalmol. 2002;120:1653–8. [PubMed] [Google Scholar]
26. Honavar SG. Emerging options in the management of advanced intraocular retinoblastoma. Br J Ophthalmol. 2009;93:848–9. [PubMed] [Google Scholar]
Br J Ophthalmol. 2009;93:848–9. [PubMed] [Google Scholar]
27. Gilbert C, Fielder A, Gordillo L, Quinn G, Semiglia R, Visintin P, et al. ; For the International NO-ROP Group. Characteristics of babies with severe retinopathy of prematurity in countries with low, moderate and high levels of development: Implications for screening programmes. Pediatrics. 2005;115:e518–25. [PubMed] [Google Scholar]
28. McColm JR, Fleck BW. Retinopathy of prematurity: Causation. Semin Neonatol. 2001;6:453–60. [PubMed] [Google Scholar]
29. Carlo WA, Finer NN, Walsh MC, Rich W, Gantz MG, Laptook AR, et al. Support Study Group of the Eunice Kennedy Shriver NICHD Neonatal Research Network. Target ranges of oxygen saturation in extremely preterm infants. N Engl J Med. 2010;362:1970–9. [PMC free article] [PubMed] [Google Scholar]
30. Wilkinson AR, Haines L, Head K, Fielder AR. UK retinopathy of prematurity guideline. Early Hum Dev. 2008;84:71–4. [PubMed] [Google Scholar]
31. Vinekar A, Dogra MR, Sangtam T, Narang A, Gupta A. Retinopathy of prematurity in Asian Indian babies weighing greater than 1250 grams at birth: Ten year data from a tertiary care center in a developing country. Indian J Ophthalmol. 2007;55:331–6. [PMC free article] [PubMed] [Google Scholar]
Retinopathy of prematurity in Asian Indian babies weighing greater than 1250 grams at birth: Ten year data from a tertiary care center in a developing country. Indian J Ophthalmol. 2007;55:331–6. [PMC free article] [PubMed] [Google Scholar]
32. Shah PK, Narendran V, Kalpana N, Gilbert C. Severe retinopathy of prematurity in big babies in India: History repeating itself? Indian J Pediatr. 2009;76:801–4. [PubMed] [Google Scholar]
33. Darlow BA, Gilbert C, Quinn GE, Azad R, Ells AL, Fielder A, et al. Promise and potential pitfalls of anti-VEGF drugs in retinopathy of prematurity. Br J Ophthalmol. 2009;93:986. [PubMed] [Google Scholar]
34. Rudanko SL, Laatikainen L. Visual impairment in children born at full term from 1972 through 1989 in Finland. Ophthalmology. 2004;111:2307–12. [PubMed] [Google Scholar]
35. Rudanko SL, Fellman V, Laatikainen L. Visual impairment in children born prematurely from 1972 through 1989. Ophthalmology. 2003;110:1639–45. [PubMed] [Google Scholar]
What Causes Blindness at Birth? | For Eyes
People of all ages can endure issues with their eyesight, even infants. From obstacles during pregnancy to developmental problems, the reasons behind vision loss in babies can vary.
From obstacles during pregnancy to developmental problems, the reasons behind vision loss in babies can vary.
To find out more about what causes blindness at birth, keep reading. For Eyes is here to help.
What causes blindness at birth?
Like other age groups, newborns are not immune to blindness. Your little one’s eyes will need to be inspected from the time they’re born, such as for congenital cataracts, which may appear as unpigmented spots on your child’s eyes.
Congenital cataracts are a potential cause of vision loss in infants that can sometimes go undetected until your child is older, which is why every child needs an eye exam by 6 months of age. (The next eye exam should be at 3 years old and then again at age 6, with an appointment every year after into adulthood.)
What else causes blindness at birth? Issues with your child’s eye development or problems with the portion of the brain that controls vision may impact your child’s eyesight.
Another one of the potential causes of blindness in infants is retinopathy of prematurity (ROP), which affects the blood vessels in the retina. It most often impacts premature babies who are less than three pounds. When an infant is born early, their retina and blood vessels may not finish developing, which can inhibit oxygen absorption around the retina.
It most often impacts premature babies who are less than three pounds. When an infant is born early, their retina and blood vessels may not finish developing, which can inhibit oxygen absorption around the retina.
Due to this, the retina may be alerted to create new blood vessels. These can be delicate, and they tend to leak fluids, which can create marks and scars. The scars can yank on the retina as your child grows, which can make the retina move from its place behind the eye. Even if retinal detachment does not occur, your little one’s vision can still be impacted, usually making their direct line of sight blurry at the least.
According to the American Optometric Association (AOA), if your child has other health issues, they may have a higher chance of developing ROP. However, how much your little one weighs and when they’re born are some of the most significant risk factors.
What are the symptoms of blindness in babies?
From the day your child is born, it’s a good idea to watch for symptoms of blindness. In babies, the way to do so is to work with an eye doctor, who will inspect how your child’s eyes function together and apart. They may also look at how your child’s eyes respond to light, which can indicate a vision concern if the light doesn’t seem to affect them.
In babies, the way to do so is to work with an eye doctor, who will inspect how your child’s eyes function together and apart. They may also look at how your child’s eyes respond to light, which can indicate a vision concern if the light doesn’t seem to affect them.
Here are some other common symptoms of blindness in babies and children:
- Discolored pupils
- Eye movement issues
- Light sensitivity
- Red eyes
- Touching the eyes often
Remember: These symptoms don’t always indicate oncoming blindness. When your little one is young, it can take time for their eyes to develop. If you notice any symptoms happening more often than usual, consult an Independent Doctor of Optometry in your area.
Summary: Blindness in Babies
Monitoring symptoms of blindness in babies and children is the best way to detect eye issues early on and get your child the right treatment. If your child exhibits any signs of eye issues, such as discoloration in the pupils or light sensitivity, we encourage you to reach out to an optometrist.
Are you curious about what causes blindness at birth? Two of the main conditions to watch for are congenital cataracts and ROP.
While these issues are by no means the only ones that can affect your baby’s eyes, they’re some important ones to monitor. By ensuring your little one gets a comprehensive eye exam by 6 months old, you can usually catch these conditions before they progress too much.
Book your eye exam at For Eyes
Have you had your annual comprehensive eye exam? Schedule an appointment with an Independent Doctor of Optometry at your local For Eyes.
Book an Exam
Kids Eye Health Eyeglasses eye doctor eye exam Eye Health eye pain question eye doctor symptoms eye eye eyewear routine eye exam birth BlindnessThere are more and more blind children.
 As he noted, this problem is one of the most urgent in medicine. A blind child is born every minute in the world. According to WHO, about 1.5 million children are currently blind. In 2000, the global economic cost of blindness was $19 billion. However, 80% of cases of blindness can be avoided. nine0004
As he noted, this problem is one of the most urgent in medicine. A blind child is born every minute in the world. According to WHO, about 1.5 million children are currently blind. In 2000, the global economic cost of blindness was $19 billion. However, 80% of cases of blindness can be avoided. nine0004 In Russia, blindness after retinopathy of prematurity remains the highest in the world, said E. Sidorenko. Every year, about 5,000 children become visually impaired (for comparison: in the USA there are about 500), and only 5% undergo rehabilitation. Russia confidently comes out on top in terms of visual disability: for example, in the 1950s, only 6% of the pupils of the blind schools were premature, in 1994 -14%, and now - more than 60% (for comparison: in the UK - 11%, in Japan - 15%). Thus, retinopathy (ophthalmopathy) of preterm infants is a severe vascular proliferative disease that continues to be one of the most pressing problems of modern ophthalmology, forming an army of the blind. nine0005
nine0005
The power of the proliferative process in retinopathy, the speed and irreversibility of its development, the lack of effective treatment often plunge pediatric ophthalmic surgeons into despondency and pessimism. The rapid transition from one stage of retinopathy to another, a large number of comorbidities, the severe general condition of very premature babies, which make it difficult to carry out cryo- or laser coagulation in a timely manner, often lead to a loss of time and the development of a cicatricial stage of the process. In this case, a powerful retrolental film is formed, total retinal detachment with severe fibrosis of the vitreous body, often in both eyes, with loss of visual functions. nine0005
The decision taken in Russia in 1992 to nurse extremely premature babies weighing more than 1000 g was inadequate in the context of the collapsing healthcare of that time. Against the backdrop of political, demographic, economic, social problems, this contributed to a significant increase in disability among preterm infants, especially visual impairment.
In the 90s in Moscow there were only 17% of normal deliveries. Every 10th child was premature with incomplete formation of the vascular tract of the eye. Fortunately, in 60% of cases it ended normally, but every 4-5th premature baby entered the path of vision loss due to retinopathy. nine0005
The use of lasers, cryoapplications made it possible to stop the development of retinopathy of prematurity in 60-80% of cases. Despite the successes achieved, many unresolved problems remain in Russia and other countries. Early morbidity and its steady growth are accompanied by an increase in the number of disabled children. Visual disability since childhood is approaching 21% of the total number of visually impaired people. In 70% of cases in adult patients, visual disability is the outcome of a disease that began in childhood. nine0005
Vision can be saved!
At the Department of Ophthalmology, Faculty of Pediatrics, Russian State Medical University. N.I. Pirogov, congenital pathology of the organ of vision in children is traditionally one of the main topics of scientific research. Along with in-depth scientific research on the clinic of this disease, work is underway to study the etiology of the process, where a special place is given to the viral problem.
Pirogov, congenital pathology of the organ of vision in children is traditionally one of the main topics of scientific research. Along with in-depth scientific research on the clinic of this disease, work is underway to study the etiology of the process, where a special place is given to the viral problem.
The first article in Russia on retinopathy of prematurity was published by the staff of the department at 1993. For the first time in Russia, the features of the eyes of premature babies were studied depending on the gestational age, which made it possible to differentiate the norm from pathology. This facilitated the diagnosis of retinopathy of prematurity. For the first time in world practice, the morphological structure of eye structures depending on gestational age, which provide hydrodynamic homeostasis of the eye of a premature baby, has been studied. By the 24th week of gestational age, these structures are morphologically immature, which leads to low intraocular pressure in the eye. These studies are of great importance for the detection of congenital glaucoma and the prevention of blindness. nine0005
These studies are of great importance for the detection of congenital glaucoma and the prevention of blindness. nine0005
The study of the pathogenesis of retinopathy of prematurity allowed the staff of the department for the first time in the world to develop a method for preventing the development of retinopathy of prematurity. Special equipment has been created, a special eye chamber, where a variable vacuum pressure is created above the child's eye. An alternating vacuum is used to create blood flow from the posterior segment of the eye to the avascular zone. Variable exposure avoids vascular damage, improves hemo- and hydrodynamics and eliminates circular hypoxia. A new technique has been developed, which made it possible to reduce the risk of developing blindness after this disease by 2 times. nine0005
Conducted for the first time in the world practice, studies of the development of autoregulation of eye hemodynamics in premature infants made it possible to establish that hypoxic and hypercapnic drives of biochemical autoregulation of the eye in premature infants have not yet been formed. The hypoxic drive works more clearly, causing inadequate angiospasm of the retinal vessels and the development of circulatory hypoxia. The hypercapnic drive matures only by the 29th week of gestational age (7.5 months). Accounting for these features allows you to individually select the mode of oxygen supply for each child, taking into account the reaction of the vessels of the eye. nine0027
The hypoxic drive works more clearly, causing inadequate angiospasm of the retinal vessels and the development of circulatory hypoxia. The hypercapnic drive matures only by the 29th week of gestational age (7.5 months). Accounting for these features allows you to individually select the mode of oxygen supply for each child, taking into account the reaction of the vessels of the eye. nine0027
New scientific data on the study of oxygen exchange in the vitreous body of the eye for the first time showed the accumulation of a high level of oxygen there, which significantly exceeds the partial tension of oxygen in the inhaled mixture and the blood of the child. These studies reveal new causes of the onset and progression of retinopathy of prematurity, dictate the development of new methods for correcting oxygen metabolism.
The department has developed risk factors for the development of newborn retinopathy, including ultrasound. According to foreign authors, premature babies born with a weight of less than 1.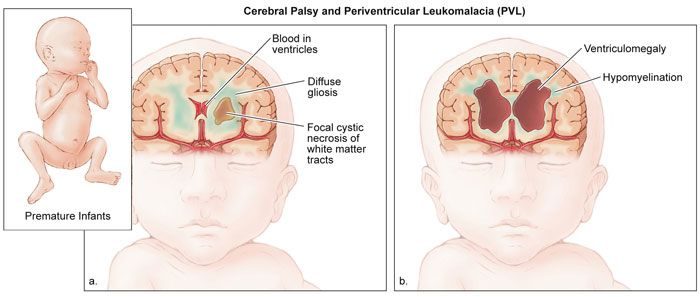 5 kg and at a gestational age of less than 32 weeks are classified as children at risk. However, in the conditions of nursing premature babies in Russia, a slightly different contingent of children belongs to the risk group - in our country, retinopathy is also found in more full-term children - those born with a weight of up to 2.5 kg and for up to 34 weeks. Neurosonographic examination in this group of children is dominated by the picture of cerebral ischemia. An increase in vascular resistance in the basin of the anterior cerebral artery was noted on the eve of development and against the background of the active course of retinopathy of prematurity. An increase in the resistance index of the anterior cerebral artery was revealed in conditions preceding the development of retinopathy. nine0005
5 kg and at a gestational age of less than 32 weeks are classified as children at risk. However, in the conditions of nursing premature babies in Russia, a slightly different contingent of children belongs to the risk group - in our country, retinopathy is also found in more full-term children - those born with a weight of up to 2.5 kg and for up to 34 weeks. Neurosonographic examination in this group of children is dominated by the picture of cerebral ischemia. An increase in vascular resistance in the basin of the anterior cerebral artery was noted on the eve of development and against the background of the active course of retinopathy of prematurity. An increase in the resistance index of the anterior cerebral artery was revealed in conditions preceding the development of retinopathy. nine0005
Currently, the department is conducting immunological studies that will facilitate the prognosis of the development of retinopathy of prematurity, determine the causes of delayed development of the disease 1-2 months after birth, and improve the method of nursing children with retinopathy of prematurity. The study of the possibility of using stem cells to improve the function of the retina will improve vision in children both after conservative and especially after surgical treatment of severe cicatricial stages of the disease. nine0005
The study of the possibility of using stem cells to improve the function of the retina will improve vision in children both after conservative and especially after surgical treatment of severe cicatricial stages of the disease. nine0005
The Department of Ophthalmology of the Pediatric Faculty of the Russian State Medical University introduced the methods of cryo- and laser coagulation of peripheral avascular zones of the retina into the clinical practice of Moscow clinics. These methods allow in 80% of cases to prevent the progression of an already existing disease and the development of severe stages of retinopathy of prematurity, leading to low vision and blindness.
Thus, to date, almost 80% of premature babies do not go blind from retinopathy of prematurity, but one in five is threatened by it. The department conducts experimental studies of changes in the hemodynamics of the eye after cryo- and laser coagulation of the retina and choroid to clarify the mechanism of action of this treatment method and increase its effectiveness. nine0005
nine0005
In children with an aggressive form of retinopathy of prematurity, an extended surgical intervention is performed, in which the lens, vitreous body, and all scarring are removed, together with a powerful laser surgical intervention. Operations carry the risk of eye loss and have small visual outcomes, but having even a small amount of vision greatly improves the life of the child and is worth the effort.
Conducted by the Department of Ophthalmology of the Pediatric Faculty of the Russian State Medical University, multifaceted scientific research on retinopathy of prematurity with a wide coverage of oxygen metabolism, hemodynamic autoregulation, immunological studies, stem cells leads to the creation of new treatment methods and the improvement of old ones. nine0005
Retinopathy of prematurity in Russia occupies a special place, being the result of many social problems, the speaker noted in conclusion. Domestic medicine has made a significant contribution to the development of this problem, which has reduced the number of blind children after retinopathy of prematurity by 80%. Further research in the field of finding the cause of retinopathy, immunological problems, stem cells create hopes for the elimination of the main cause of blindness in children - retinopathy of prematurity. nine0027 Childbirth from the 22nd week of pregnancy?
Further research in the field of finding the cause of retinopathy, immunological problems, stem cells create hopes for the elimination of the main cause of blindness in children - retinopathy of prematurity. nine0027 Childbirth from the 22nd week of pregnancy?
The discussion of E. Sidorenko's report was very sharp and emotional. Academician of the Russian Academy of Medical Sciences A. Brovkina called for the mandatory examination of newborn children by a pediatric ophthalmologist in maternity hospitals, and not a month after birth, as is customary now. During this time, not only retinopathy progresses, but also congenital retinoblastoma. If this malignant tumor is detected immediately after the birth of a child and surgical treatment is carried out, then it is possible to save not only life, but also vision. nine0005
Academician of the Russian Academy of Medical Sciences A.Strizhakov spoke out against the planned transition in 2012 of our country to the birth registration criteria recommended by WHO (gestational age from 22 weeks and birth weight from 500 g).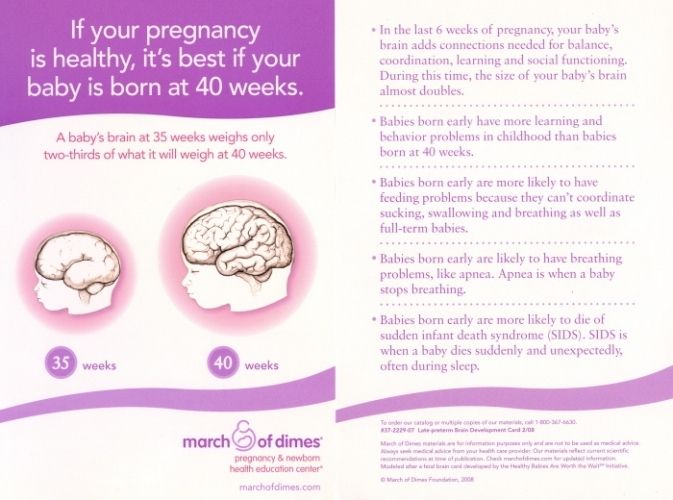 According to him, children born before 25 weeks of gestation are not only blind, but also deaf. Will we not make the same mistake as in 1992, when prematurely, with the obvious unpreparedness of the obstetric service, they began to nurse premature babies starting from the 28th week of pregnancy and weighing 1 kg? Now the equipment of most Russian perinatal centers has been brought up to the world level, but before this happened, the number of blind children increased sharply. In addition, very premature babies, in addition to severe visual pathology, have a large number of other serious diseases, including neurological ones, and we can talk about systemic disability. Even in developed countries, leading obstetricians and gynecologists call for counting the term of labor from the 25th week of pregnancy. nine0005
According to him, children born before 25 weeks of gestation are not only blind, but also deaf. Will we not make the same mistake as in 1992, when prematurely, with the obvious unpreparedness of the obstetric service, they began to nurse premature babies starting from the 28th week of pregnancy and weighing 1 kg? Now the equipment of most Russian perinatal centers has been brought up to the world level, but before this happened, the number of blind children increased sharply. In addition, very premature babies, in addition to severe visual pathology, have a large number of other serious diseases, including neurological ones, and we can talk about systemic disability. Even in developed countries, leading obstetricians and gynecologists call for counting the term of labor from the 25th week of pregnancy. nine0005
However, Academician E. Sidorenko cited Germany as an example, where a child was recently delivered at the clinic of the University of Göttingen, born with a body weight of only 275 g, who is now the smallest surviving boy in the world. After 6 months in the intensive care unit, the boy was discharged from the clinic - by this time his weight was already 3.7 kg. But European and Russian nursing of newborns are different things, President of the Russian Academy of Medical Sciences M. Davydov noted. nine0005
After 6 months in the intensive care unit, the boy was discharged from the clinic - by this time his weight was already 3.7 kg. But European and Russian nursing of newborns are different things, President of the Russian Academy of Medical Sciences M. Davydov noted. nine0005
The President of the Russian Academy of Medical Sciences touched upon another hot topic - the impact of IVF on the health of newborns, including vision. According to Academician of the Russian Academy of Medical Sciences A. Baranov, 75% of children born after IVF are sick, half of them have various eye diseases, even though they are full-term. IVF in Russia is carried out in 33 centers, of which only 3 are state-owned. “What they are doing is unknown,” said Academician of the Russian Academy of Medical Sciences A. Strizhakov. “They transplanted an egg, got money, but they don’t care how the pregnancy goes.” He urged to analyze the future of such children by conducting serious research on the impact of IVF on their health. nine0005
nine0005
According to M.Davydov, the severity and importance of the problem under discussion are obvious. So far, unfortunately, the vast majority of our maternity hospitals are not ready to nurse children born weighing less than 1 kg. In the future, they may become disabled and become a heavy burden on the shoulders of society. At a meeting of the Presidium of the Russian Academy of Medical Sciences, it was decided to prepare appropriate recommendations and send them to the Ministry of Health and Social Development of Russia and the State Duma of the Russian Federation.
Fedor SMIRNOV.
‹ Facing a global threat Up Where it is harmful and dangerous ›
Genetic predisposition to hearing loss
Every family that has a child with hearing loss asks this question. How so? Why do we, because we all hear well?
To deal with the answers to these questions, you need to delve into the theory.
Genetic predisposition
More than half of hearing loss is due to a genetic predisposition. It is the genes that determine which hereditary traits will pass from parents to children. We receive one copy of each gene from our mother and father. This genetic set can contain both normal genes and pathogenic ones that manifest as diseases. nine0005
Each of us can be a carrier of altered, pathogenic genes. And the problem is that such genes may not manifest themselves, and we may not even know about their existence.
Altered genes are also called mutations. When a child receives two of these altered genes, one from each parent, the mutation appears. The probability of coincidence of two mutational genes is about 25%.
How to detect predisposition
Diagnosis of genetic predisposition is carried out in the following ways:
- Patient's hereditary history.
In other words, this is a family tree with information about chronic diseases and how they proceeded and at what age they developed. The more detailed this list is, the better. The data obtained will allow building a clear system for the diagnosis and prevention of possible diseases.
The more detailed this list is, the better. The data obtained will allow building a clear system for the diagnosis and prevention of possible diseases. - Regular audiometric examinations.
Allows you to determine the susceptibility of hearing to sound waves of different frequencies. nine0080 Molecular genetic analysis. - Auxiliary diagnostic methods.
For example, computed tomography or magnetic resonance imaging.
Detects hereditary pathologies and analyzes the presence of modifications in genes, chromosomes and DNA.
Each person can be a carrier of the gene for recessive non-syndromic deafness, which does not manifest itself in any way. Most often, people do not even know that such a gene is in their body
What to look out for
It is not always necessary to delve into the causes of what happened. Perhaps you will find out what caused and led to hearing loss in a child. And perhaps the reason will remain a mystery. Genetics is just statistics. You can't fix her. Of course, I want to know and understand how it was possible to avoid deafness. However, when there is already a problem, it is more important to raise another question: “How to help the baby?”
Genetics is just statistics. You can't fix her. Of course, I want to know and understand how it was possible to avoid deafness. However, when there is already a problem, it is more important to raise another question: “How to help the baby?”
Often, hearing loss in a baby is perceived tragically by parents. Experiencing deep mental suffering can prevent you from seeing the most important thing - your child. And for him, hearing loss is not grief at all. With timely access to specialists, the child becomes a full member of society and develops in the same way as everyone else. Sometimes even better. nine0005
Case report: boy, cochlear implants in both ears. The first - at 2 years old, the second - at 4. At the age of 14, he came from Chechnya with his father for another tuning session. In terms of development, the boy is in no way inferior to his peers. Participates in physics olympiads, knows several languages: Russian, Chechen, English. Nevertheless, at the end of the visit, the father asks the doctor: “How is it? Why are we unlucky?"
But the boy is doing well! He has two arms, two legs and two eyes. He has a family. The child lives a full life and hears. Modern hearing aids and cochlear implants can correct almost any hearing loss and enable you to enjoy life to the fullest. nine0005
He has a family. The child lives a full life and hears. Modern hearing aids and cochlear implants can correct almost any hearing loss and enable you to enjoy life to the fullest. nine0005
When making a diagnosis, the most important thing for parents is to remember the child himself and strive to help him develop and compensate for hearing
Unfortunately, not all parents accept the diagnosis of their children and the treatment plan offered by specialists. They are ready to use dubious methods without guarantee of any result: reflexology, acupuncture, drops and pills. However, all these methods are not able to compensate for hearing. They only take time that can be used for development. nine0005
Physical hearing is compensated by hearing aids. If necessary, cochlear implants. However, while waiting for surgery on the second ear, it is still advisable to use a hearing aid on it.
For full development, a child needs to hear, understand speech and speak for himself.